Method Validation Software for Clinical Laboratories and IVD Manufacturers
The complete, validated implementation of 11 CLSI protocols — measurement system analysis, method comparison, reference intervals, and diagnostic performance, all in a single package within Excel.
Method Validation Software for Clinical Laboratories and IVD Manufacturers
The complete, validated implementation of 11 CLSI protocols — measurement system analysis, method comparison, reference intervals, and diagnostic performance, all in a single package within Excel.
Try free for 15 days — no credit card required

Trusted by 75,000 scientists at most of the top-10 IVD manufacturers, and at thousands of ISO 15189, ISO/IEC 17025, and CLIA-regulated laboratories worldwide for over 30 years.






Complete CLSI method validation coverage
11 CLSI protocols across the full validation workflow — precision, bias, linearity, detection capability, method comparison, reference intervals, and diagnostic performance. Everything your laboratory verification or IVD submission requires, in one package.
Results you can stand behind
Every calculation runs in Analyse-it’s own validated engine — no Excel formulas, no third-party functions. Validated against CLSI reference datasets before every release. Defensible in a 510(k), CE-IVD submission, CAP inspection, or ISO 15189 audit.
Your data never leaves your environment
Analyse-it runs entirely on your PC. Nothing is sent to a server, nothing is exposed to a cloud service - important when working with pre-submission or patient-adjacent data under data governance constraints.
Method validation covers a lot of ground. IVD manufacturers need to establish precision, linearity, bias, and detection capability during development; demonstrate method comparability; and establish reference intervals and diagnostic accuracy for product labelling and regulatory submissions. Laboratories need to verify they can meet manufacturer claims before a new measurement system goes into clinical use, and demonstrate continued compliance under CLIA, CAP, ISO 15189, or ISO/IEC 17025.
Most tools cover part of this. EP Evaluator covers some CLSI protocols but not all, and requires manual data entry. Standalone statistical packages cover the statistics but not the CLSI workflows. Spreadsheet templates handle the basics but can’t be defended in a regulatory submission. Analyse-it covers all 11 CLSI method validation protocols, in a single validated package, inside Excel — so you can import data directly from instruments, run the analysis, and generate publication-ready reports without leaving the environment your team already works in.
Every calculation is performed by Analyse-it’s own engine — not in Excel formulas that can silently break when a cell reference changes. Results are validated against the example datasets published in the CLSI guidelines before every release.
Scientists at Abbott, Roche, Thermo Fisher, and most of the world’s leading IVD manufacturers have relied on it for over 30 years — as have laboratory directors and scientists at thousands of accredited and regulated laboratories worldwide.
Seen in the field
What's included
-
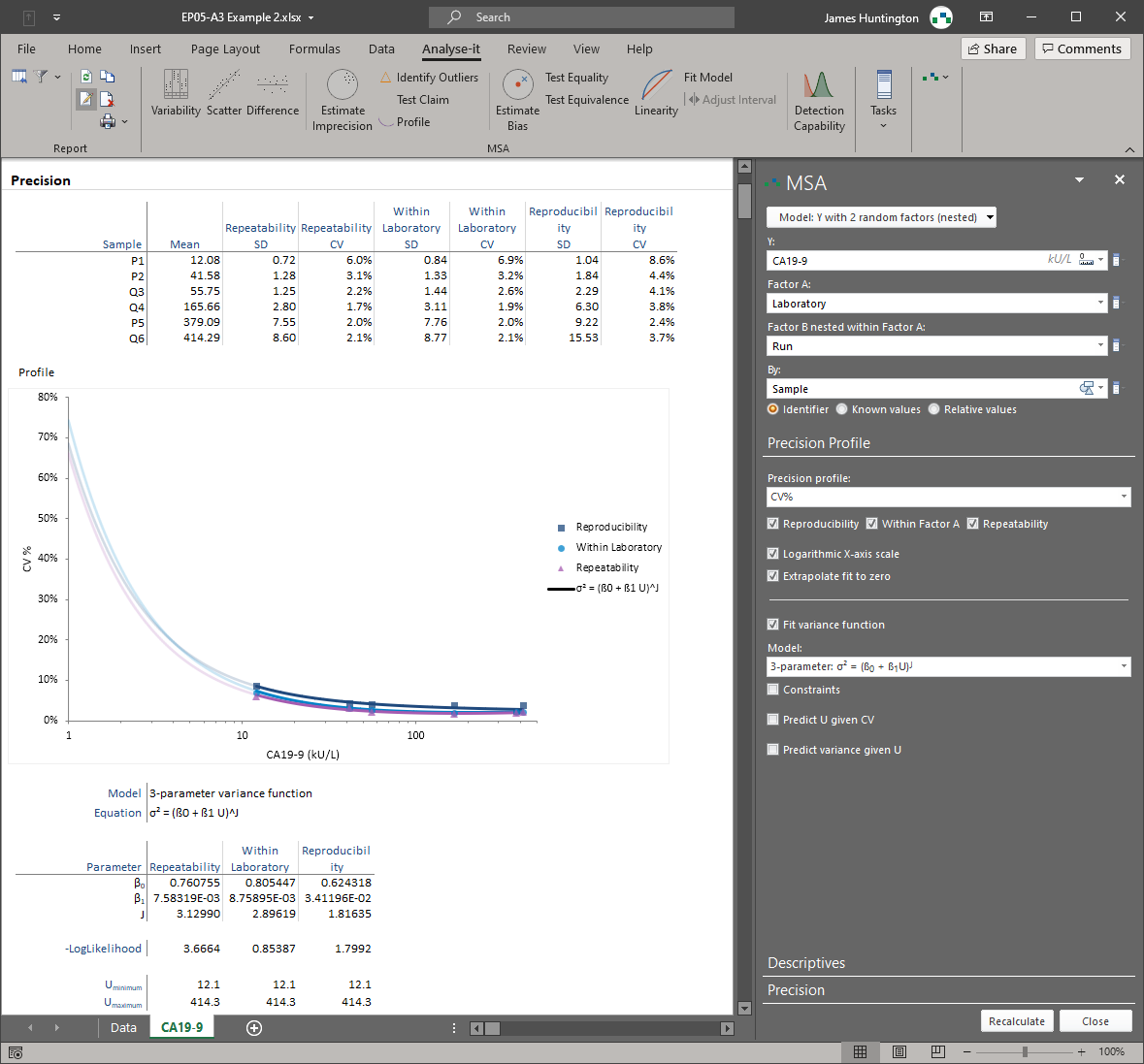
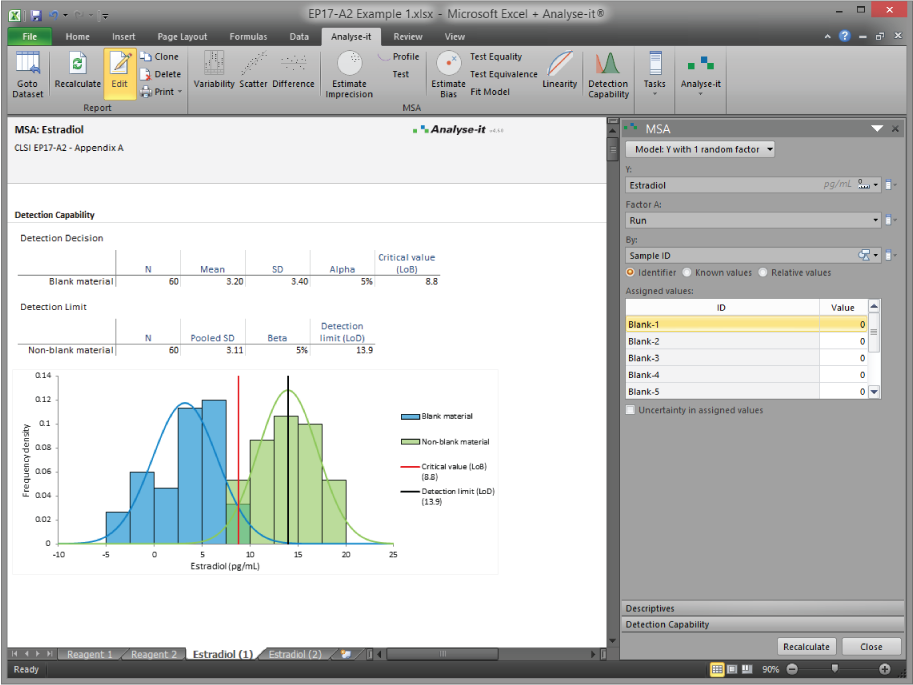
Measurement system analysis — EP05, EP06, EP10, EP15, EP17
Establish and verify precision (EP05-A3), linearity (EP06-A), bias and trueness (EP15-A3), detection capability (EP17-A2), and preliminary performance characteristics (EP10-A3). Flexible study designs without fixed run/day constraints. Precision profiles, variance function fitting, LoB, LoD, and LoQ estimation. Everything needed to characterise a measurement system from early development through to regulatory submission and field verification.
-
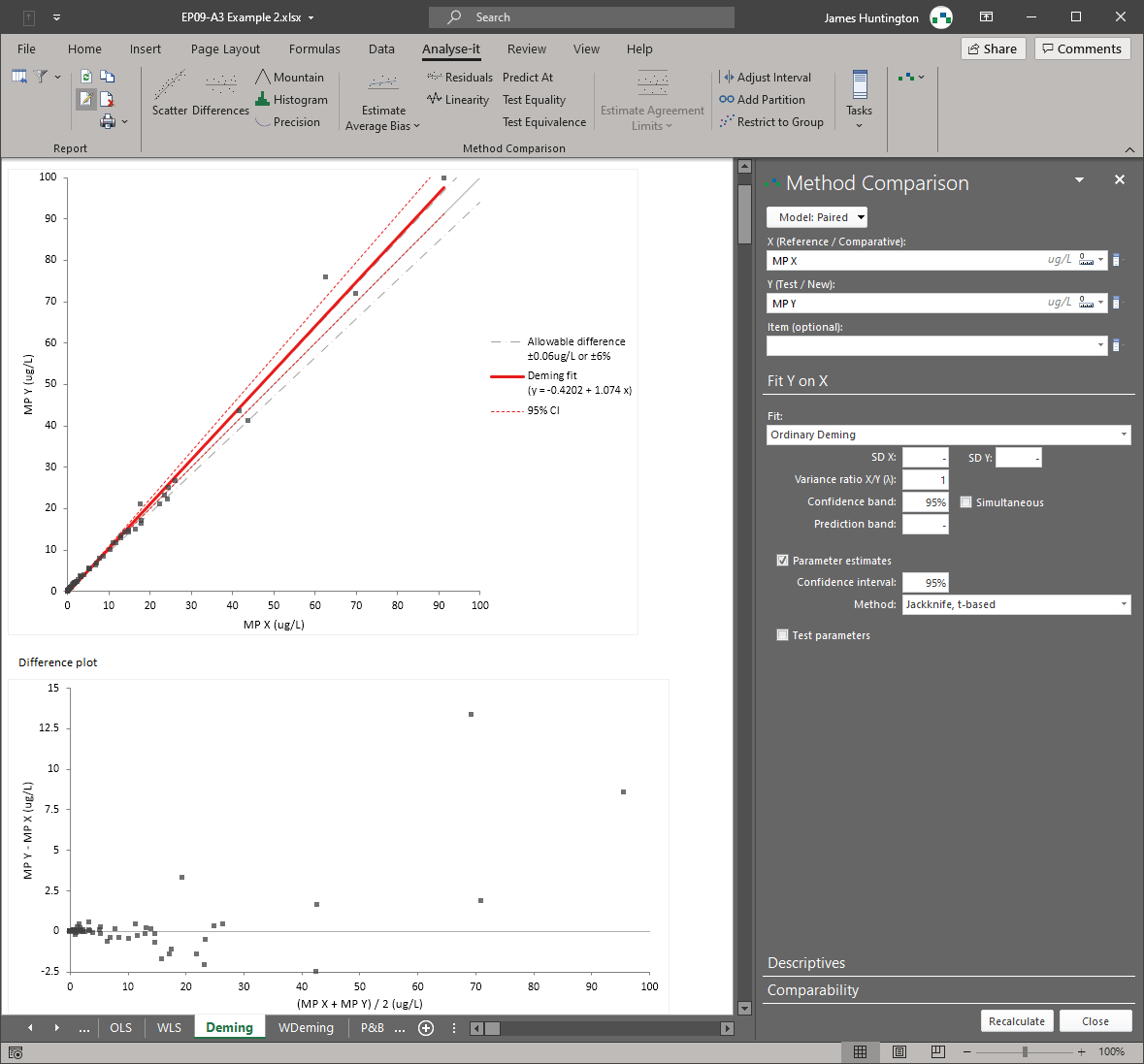
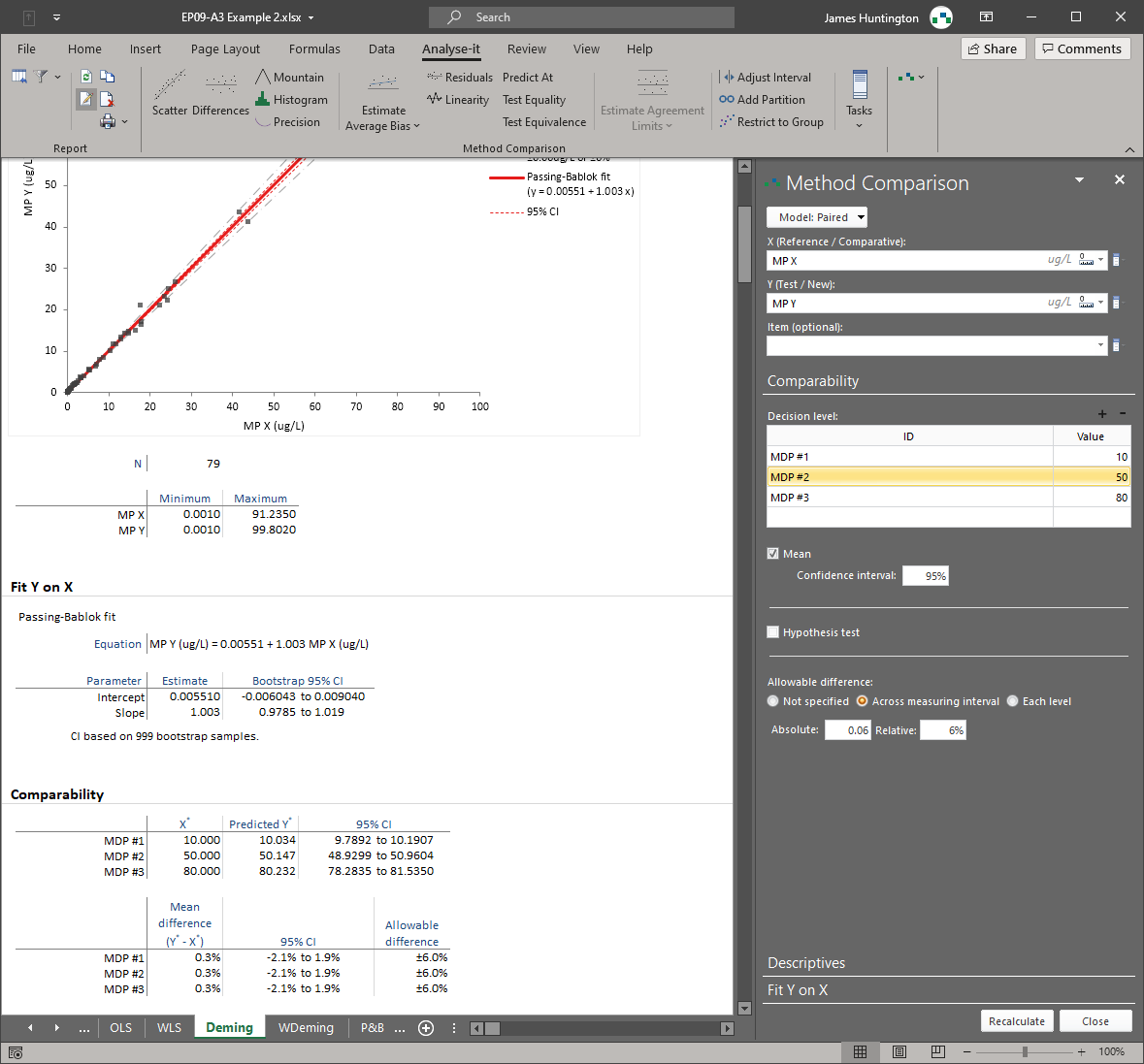
Method comparison — EP09, EP21
Bland–Altman limits of agreement, Passing–Bablok, Deming, Weighted Deming, OLS, and Weighted OLS regression — all six methods in a single analysis. Bias at clinical decision points, replicate measurement support, interval partitioning, and total analytical error estimation (EP21). For instrument verification, method changeover, and regulatory comparability studies.
-
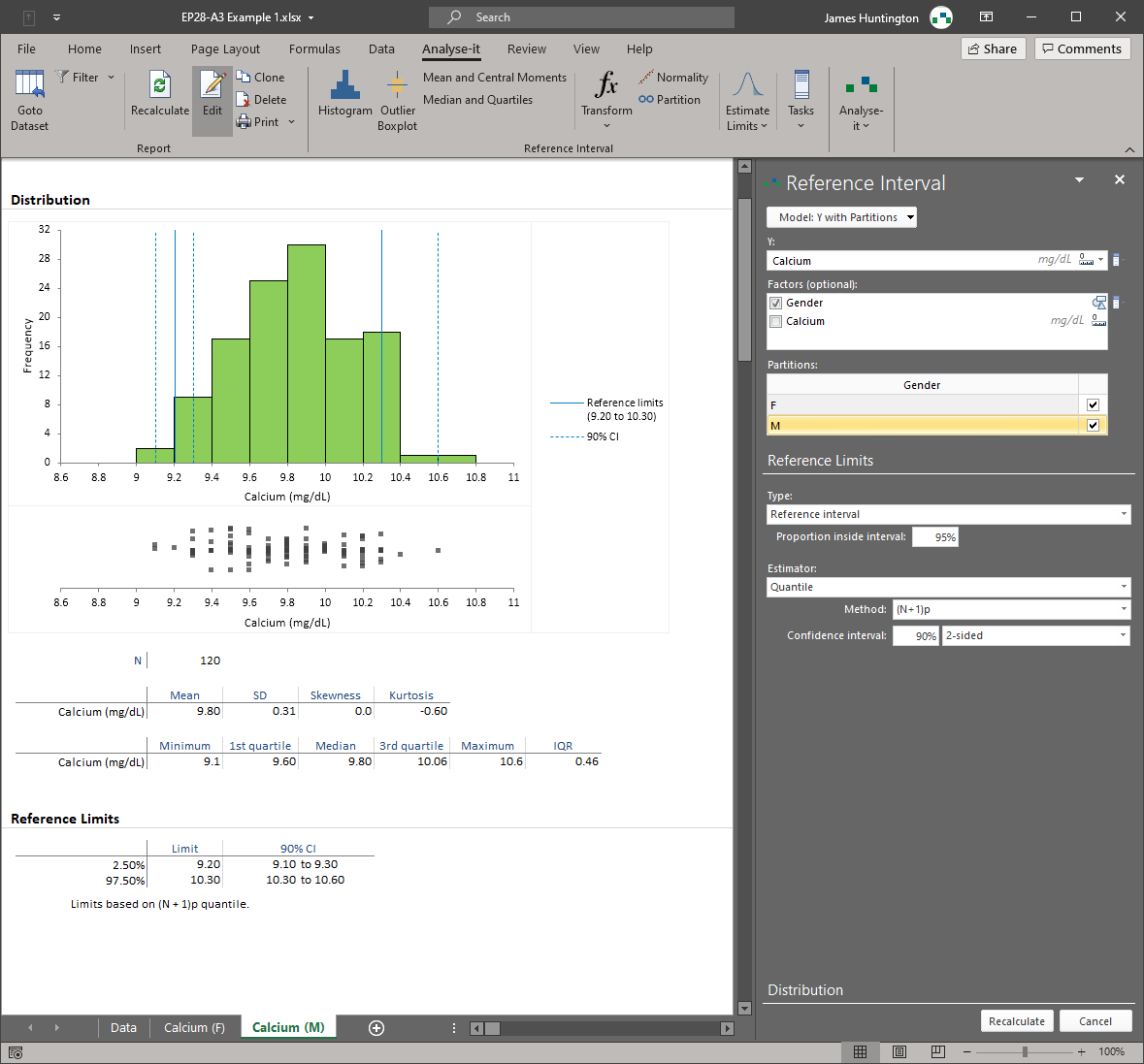
Reference intervals — EP28
Establish reference intervals using parametric, non-parametric, robust, bootstrap, and Harrell-Davis quantile methods. Partition by sex, age, ethnicity, or any clinically relevant factor. Full range of transformations for non-normal data. Transfer existing intervals to a new measurement procedure or verify that manufacturer-provided intervals are applicable to your patient population.
-
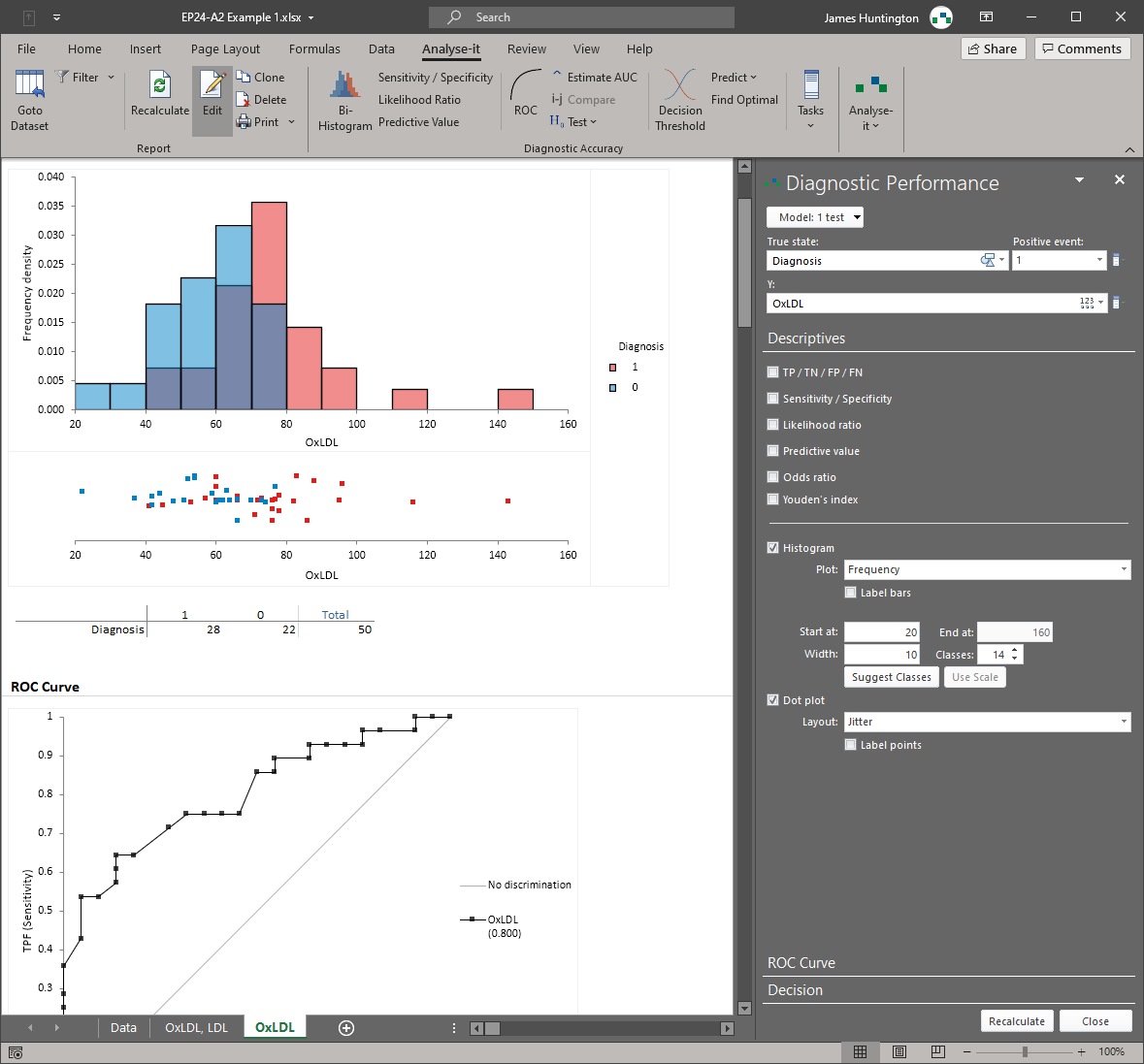
Diagnostic performance — EP24, EP12
ROC curve analysis for quantitative tests (EP24-A2) and qualitative test evaluation (EP12-A2). Sensitivity, specificity, AUC with confidence intervals, test comparison using DeLong methods, optimal threshold determination by Youden index or cost-based optimisation, and subgroup analysis. Rated best ROC curve software — Clinical Chemistry, March 2003.
-
Publication-ready reports
Generate clean, formatted reports directly from every analysis. No reformatting, no copy-paste errors, no additional tools required — whether the output is going into a regulatory dossier, a 510(k) submission, a laboratory validation file, or a peer-reviewed publication.
-
Validated before every release
Every calculation across all 11 CLSI protocols is performed by Analyse-it’s own engine — no Excel formulas or functions. Results are validated against the example datasets in the relevant CLSI guidelines before every release, so you can trust what you’re signing off on. See our development and validation process →
-
Integrated into Microsoft Excel
Analyse-it integrates directly into Excel 2007 through to Microsoft 365. Import data from instruments, analyse it immediately, and generate reports without learning new software or managing proprietary file formats. All data and results stay in Excel workbooks, making it straightforward to collaborate and share with colleagues.
-
Your data never leaves your environment
Analyse-it runs locally on your PC. No cloud processing, no data transmission, no exposure risk — your pre-submission and instrument data stays where it belongs.
-
We use Analyse-it frequently for our verification and pre-verification work, in accordance with CLSI guidelines for in-vitro diagnostics. It's saved time and effort compared to the hodge-podge of applications we used before, JMP, SAS, etc...Brian Noland, Ph.D.Principal Scientist, Product DevelopmentBiosite / Inverness Medical Innovations
-
We use Analyse-it for the analysis of data necessary to file 510k. We chose Analyse-it because it works in Excel, includes CLSI protocols, and, unlike EP-Evaluator, lets us analyze data directly from equipment without typing.Thomas D Harrigan, Ph.D.Technical Product ManagerAlfa Wassermann Diagnostic Technologies
-
I used Analyse-It for many product development, product troubleshooting, and technology evaluation activities... your product was the easiest to use, was accurate, and produced publication ready reports.Stanley F. Cernosek, Ph.D.Clinical Chemistry Reagent DevelopmentBeckman Coulter, Inc.
-
Analyse-it has been a tremendous help. I've published and presented at national cardiology meetings and couldn't have accomplished most of my research without it. Using Analyse-it, I even found errors or omissions in the work of our statistician!Regina S. Druz, MD, FACC, FASNCDirector, Nuclear CardiologyNorth Shore University Hospital
-
Although we only scratch the surface of Analyse-it’s capabilities, we have a very high volume of use for the statistics we need. It’s saved us time and the reports look professional.Michael SavageChemistry SupervisorBaptist Hospital East
Join 75,000 scientists who rely on Analyse-it. No credit card required. Full functionality from day one.
Try free for 15 days — no credit card required