Measurement system analysis software per CLSI EP05, EP06, EP15, and EP17 Precision, trueness, linearity, interferences, and detection capability — five CLSI protocols in a single validated package. Flexible nested designs for multi-site studies, verification against manufacturer claims, and LoB/LoD estimation.
Try free for 15 days — no credit card required

Trusted by 75,000 scientists at most of the top-10 IVD manufacturers, and at thousands of ISO 15189, ISO/IEC 17025, and CLIA-regulated laboratories worldwide for over 30 years.






Five CLSI protocols, one workflow
Precision (EP05-A3), linearity (EP06-A), interferences (EP10-A3-AMD), verification (EP15-A3), and detection capability (EP17-A2) in a single package. Run the full measurement system characterisation without switching tools, re-importing data, or manually configuring each analysis.
Validated against CLSI reference data
Every calculation runs in Analyse-it's own engine — no Excel formulas, no third-party functions. Results are validated against the example datasets published in the CLSI guidelines before every release. Defensible in a 510(k), CE-IVD technical file, CAP inspection, or ISO 15189 audit.
Data stays in your facility
Analyse-it runs entirely within Microsoft Excel on your PC. No cloud processing, no data transmission — your pre-submission and instrument performance data stays within your facility under your data governance controls.
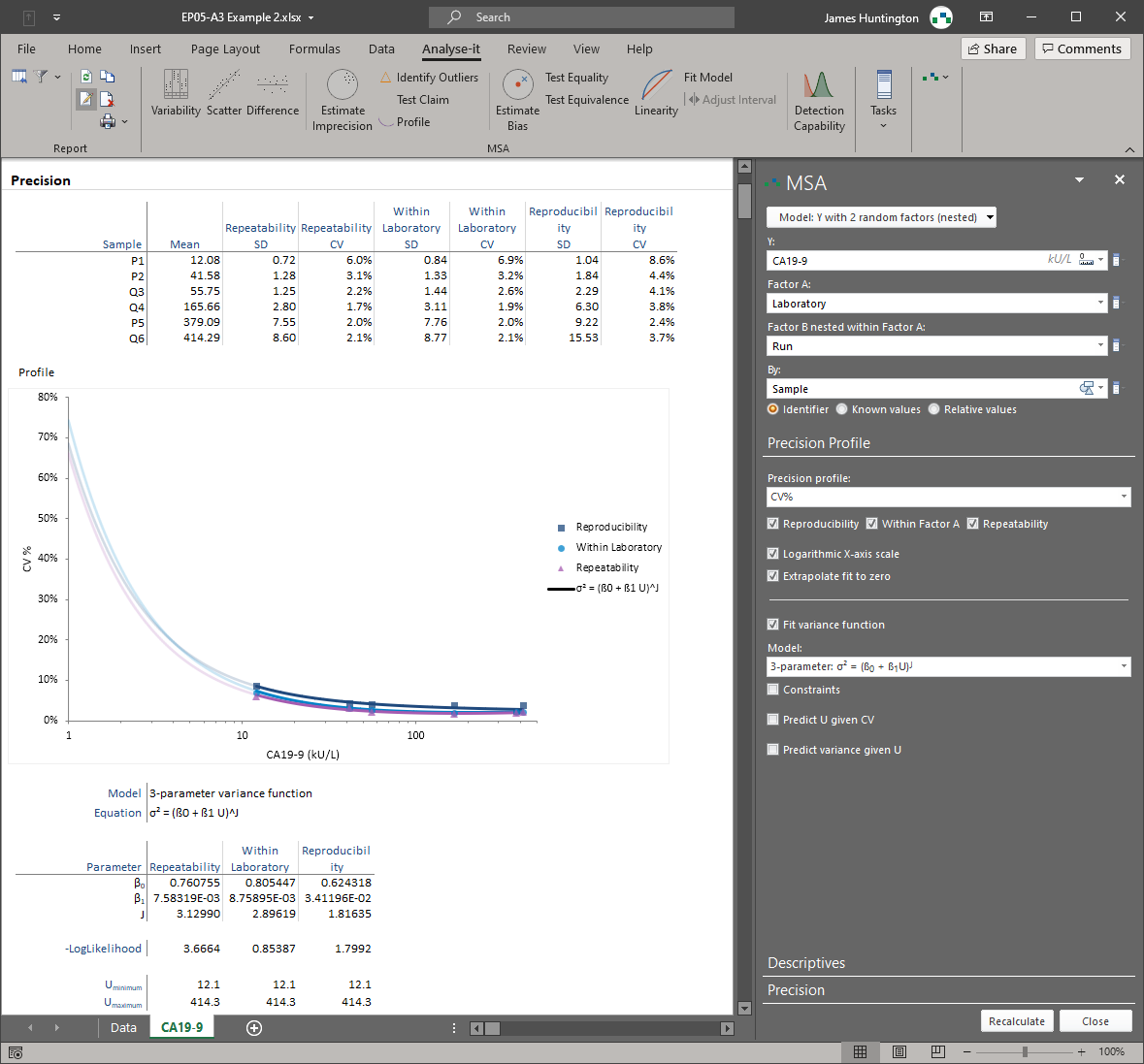
Characterising a measurement system requires multiple studies, each governed by its own CLSI protocol, each with its own experimental design, and each producing results that feed into a broader picture of system performance. Precision studies follow EP05-A3 with nested variance component analysis across runs, days, sites, and operators. Linearity studies follow EP06-A with polynomial regression and a nonlinearity test against a performance goal. Verification follows EP15-A3 by testing imprecision and trueness against manufacturer claims. Detection capability follows EP17-A2 with LoB and LoD estimation using blank and low-level materials. Most tools cover one or two of these protocols; Analyse-it covers all five in a single package, so the entire characterisation lives in one place.
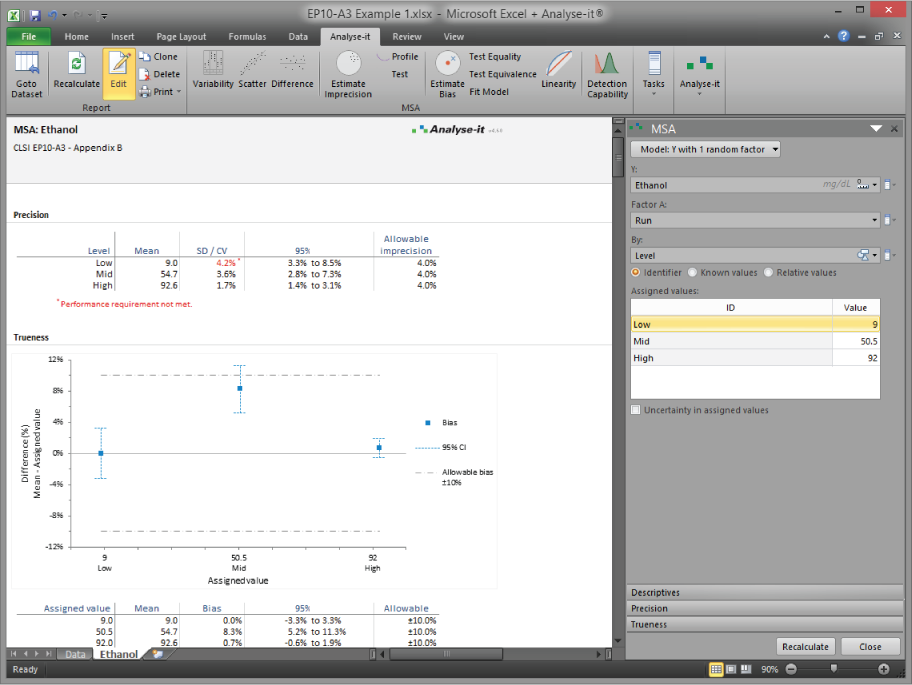
Analyse-it's precision study design is more flexible than most implementations. EP05-A3 specifies nested variance components, but it does not mandate a fixed run-within-day-within-site structure. Analyse-it supports up to three nested factors in any arrangement — runs within days, days within sites, operators within instruments — so you can design the study that matches your actual laboratory workflow rather than forcing your workflow into a rigid template. Variance components are estimated correctly for the design you specify, with repeatability, between-run, between-day, and within-laboratory imprecision reported separately. Verification per EP15-A3 then tests these estimates against the manufacturer's claimed precision and trueness, producing a clear pass/fail against the allowable goal.
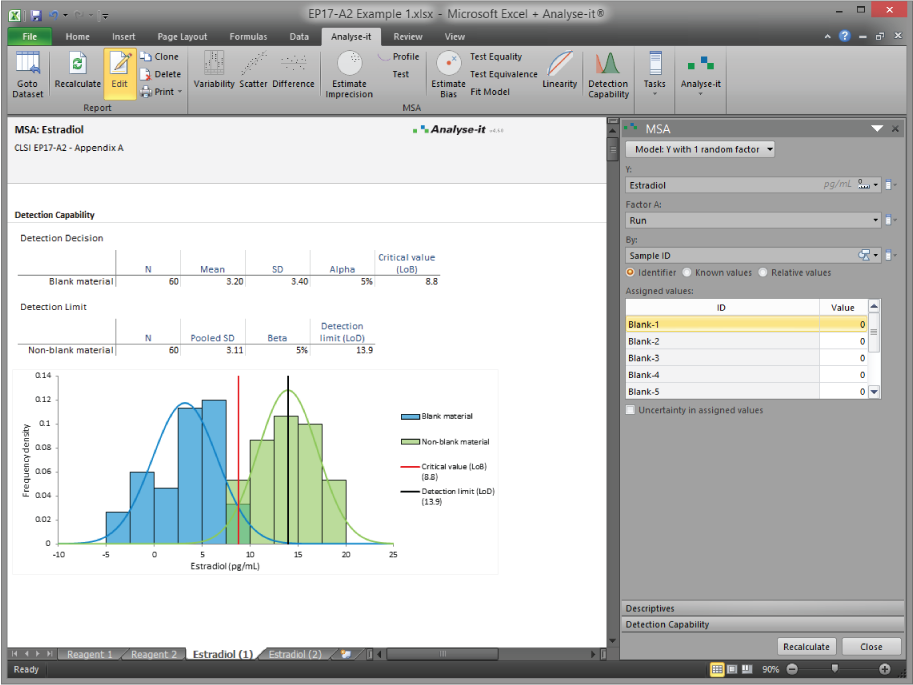
Linearity evaluation per EP06-A fits polynomial models across the measuring interval and tests for nonlinearity against a predefined performance goal. If nonlinearity exceeds the allowable deviation, the reportable range may need to be restricted — the analysis tells you where and by how much. For detection capability, EP17-A2 estimation of LoB, LoD, and LoQ uses blank and low-level sample measurements, with both classical and precision-profile-based approaches available. Interference testing per EP10-A3-AMD identifies and quantifies the effect of potentially interfering substances on measurement performance.
Scientists at Abbott, Roche, Thermo Fisher, and most of the world's leading IVD manufacturers have relied on Analyse-it for measurement system characterisation for over 30 years — as have clinical laboratories running verification studies before putting a new analyser or reagent system into clinical use.
Seen in the field
What's included
-
Establish precision with flexible nested designs (EP05-A3)
Variance component analysis with up to three nested factors in any arrangement — runs within days, days within sites, operators within instruments. Repeatability, between-run, between-day, and within-laboratory imprecision reported separately. Not limited to the standard run-within-day template.
-
Verify manufacturer claims for precision and trueness (EP15-A3)
Test observed imprecision and trueness against the manufacturer's claimed performance or an allowable analytical goal. Clear pass/fail verification with statistical hypothesis tests, not just visual comparison.
-
Evaluate linearity across the measuring interval (EP06-A)
Polynomial regression across the measuring range with a formal nonlinearity test against a predefined performance goal. Determine where and by how much the measurement system deviates from linearity, and whether the reportable range needs to be restricted.
-
Estimate LoB, LoD, and LoQ (EP17-A2)
Limit of blank and limit of detection using blank and low-level sample measurements per CLSI EP17-A2. Both classical and precision-profile-based approaches. Limit of quantitation estimation for assays where measurement uncertainty at low concentrations determines the clinically reportable range.
-
Identify and quantify interferences (EP10-A3-AMD)
Evaluate the effect of potentially interfering substances on measurement performance. Estimate the magnitude of the interference and determine whether it exceeds an allowable limit at clinically relevant concentrations.
-
Validated engine you can defend at inspection
Every calculation runs in Analyse-it's own engine — no Excel formulas or third-party functions. Validated against the CLSI published example datasets before every release. See the development and validation process.
-
Part of the Method Validation edition
Measurement system analysis is included in the Method Validation edition, which covers the full CLSI method validation workflow — method comparison (EP09-A3, EP21-A), reference intervals (EP28-A3C), diagnostic performance (EP24-A2, EP12-A2), and total analytical error (EP21-A). One tool for everything your laboratory or IVD submission requires.
-
We use Analyse-it frequently for our verification and pre-verification work, in accordance with CLSI guidelines for in-vitro diagnostics. It's saved time and effort compared to the hodge-podge of applications we used before, JMP, SAS, etc...Brian Noland, Ph.D.Principal Scientist, Product DevelopmentBiosite / Inverness Medical Innovations
-
We use Analyse-it for the analysis of data necessary to file 510k. We chose Analyse-it because it works in Excel, includes CLSI protocols, and, unlike EP-Evaluator, lets us analyze data directly from equipment without typing.Thomas D Harrigan, Ph.D.Technical Product ManagerAlfa Wassermann Diagnostic Technologies
-
I used Analyse-It for many product development, product troubleshooting, and technology evaluation activities... your product was the easiest to use, was accurate, and produced publication ready reports.Stanley F. Cernosek, Ph.D.Clinical Chemistry Reagent DevelopmentBeckman Coulter, Inc.
-
Analyse-it has been a tremendous help. I've published and presented at national cardiology meetings and couldn't have accomplished most of my research without it. Using Analyse-it, I even found errors or omissions in the work of our statistician!Regina S. Druz, MD, FACC, FASNCDirector, Nuclear CardiologyNorth Shore University Hospital
-
Although we only scratch the surface of Analyse-it’s capabilities, we have a very high volume of use for the statistics we need. It’s saved us time and the reports look professional.Michael SavageChemistry SupervisorBaptist Hospital East
Trusted by 75,000 scientists at most of the top-10 IVD manufacturers, and at thousands of ISO 15189, ISO/IEC 17025, and CLIA-regulated laboratories worldwide for over 30 years.
Try free for 15 days — no credit card required
See all Method Validation features
Pricing and licensing
Reference lab case study