Method validation software used in regulatory submissions worldwide From method comparison and precision through to linearity, reference intervals, and detection limits — 11 CLSI protocols in a single validated package, for IVD manufacturers, clinical laboratories, and regulatory professionals.
Trusted by 75,000 scientists at thousands of laboratories and 8 of the top-10 leading IVD manufacturers for over 30 years.








Technical Product Manager
Alfa Wassermann Diagnostic Technologies
Analyse-it is developed for and is in use at thousands of ISO/IEC 17025 accredited testing and calibration laboratories, ISO 15189 accredited medical laboratories, CLIA ’88 regulated medical laboratories, and IVD manufacturers for development, support, product labelling and FDA 510(k) submissions.
Establish precision, linearity, bias, detection capability, and interferences during product development. Compare your method against a predicate or reference procedure. Establish reference intervals and diagnostic accuracy for product labelling. Verify manufacturer claims when bringing a new analyser or reagent system into clinical use. Produce the statistical evidence for a 510(k) submission, CE-IVD technical file, CAP inspection, or ISO 15189 audit.
Characterise measurement system performance
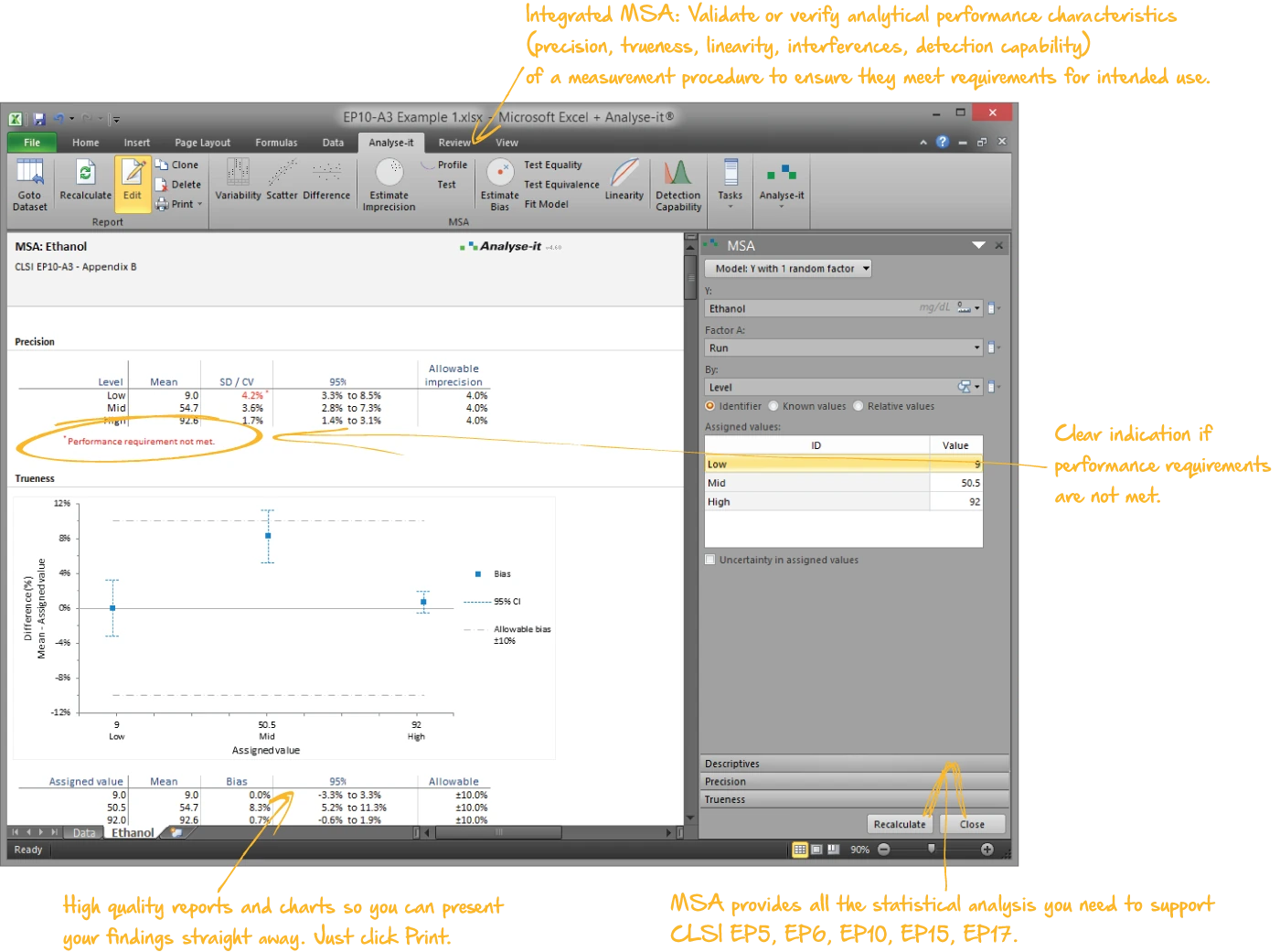
Establish the precision, trueness, linearity, interferences, and detection capability of a measurement procedure — whether you’re an IVD manufacturer characterising performance during development for a 510(k) submission, or a clinical laboratory verifying manufacturer claims:
- Precision with flexible nested variance component designs — repeatability, between-run, between-day, between-operator, between-site, within-laboratory, and reproducibility (EP05-A3)
- Precision profiles with six variance function models
- Linearity with polynomial regression, Hsieh-Liu nonlinearity testing, and adjustable measuring interval for reportable range determination (EP06-A)
- Detection capability — LoB, LoD, and LoQ using parametric, non-parametric, probit regression, and variance function approaches (EP17-A2)
- Verification of precision and trueness against manufacturer claims — such as testing observed CV against a claimed 3.5% — with χ² and equivalence tests (EP15-A3)
- Interference testing against allowable limits at clinically relevant concentrations (EP10-A3-AMD)
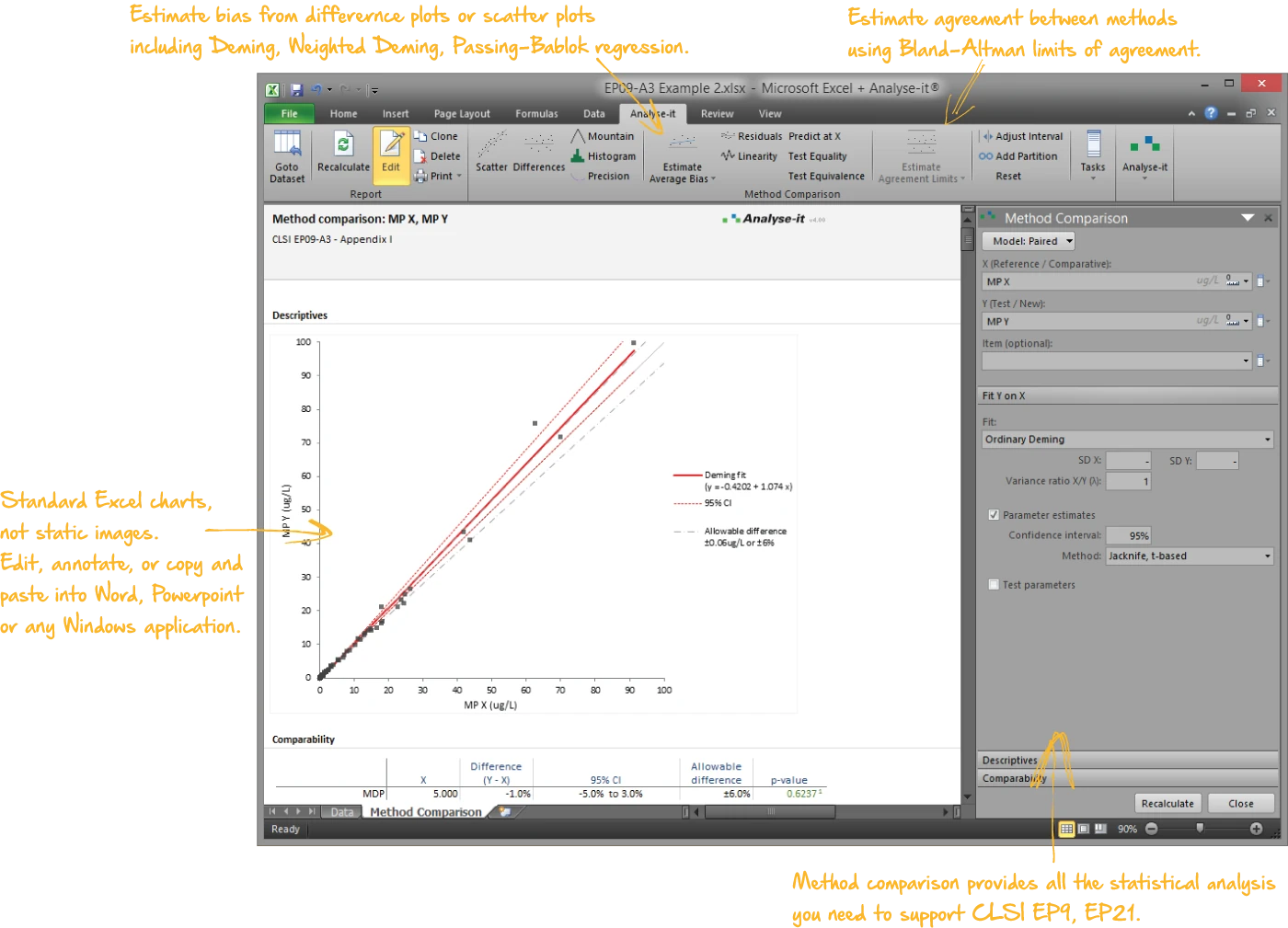
Compare methods and estimate bias
When introducing a new measurement procedure, replacing an existing one, or comparing against a reference method, you need to know the bias and whether it affects clinical decisions:
- Passing-Bablok regression (1983 and 1988) — non-parametric, robust to outliers, with CUSUM linearity test and bootstrap CIs
- Deming and Weighted Deming regression — accounts for measurement error in both methods, systematic error decomposition into constant and proportional bias, Syx independent precision estimate
- Bland-Altman limits of agreement — mean and median bias, constant and non-constant precision, linear fit for concentration-dependent bias, allowable difference band
- Bias at specified clinical decision points with confidence intervals, equality and equivalence tests. Allowable difference as absolute, percentage, or combination — such as “10%, with a minimum of 5 mg/dL”
- Total analytical error per EP21-A — combine bias + imprecision against allowable total error
- Partition the measuring range into separate intervals, each with its own fit and allowable difference specification
- Commutability per EP14-A3 — compare commutability of processed samples with prediction intervals
Establish and verify reference intervals
Reference intervals are essential for clinicians to interpret results and make a diagnosis. Analyse-it covers the full EP28-A3C workflow — from choosing the right quantile method for your data and sample size, through outlier screening and partitioning, to transfer and verification against a new procedure or population.
- Five quantile methods — parametric, non-parametric (three computation approaches), robust bi-weight, bootstrap, and Harrell-Davis
- Partition by factors that affect the intervals — such as sex and age group for alkaline phosphatase
- Full range of transformations — log, square root, cube root, Box-Cox, Manly exponential, two-stage exponential/modulus
- Outliers with Tukey box plots, and normality assessment with Shapiro-Wilk, Anderson-Darling and frequency histogram
- Transfer and verify intervals — typically 20 samples per partition per EP28-A3C
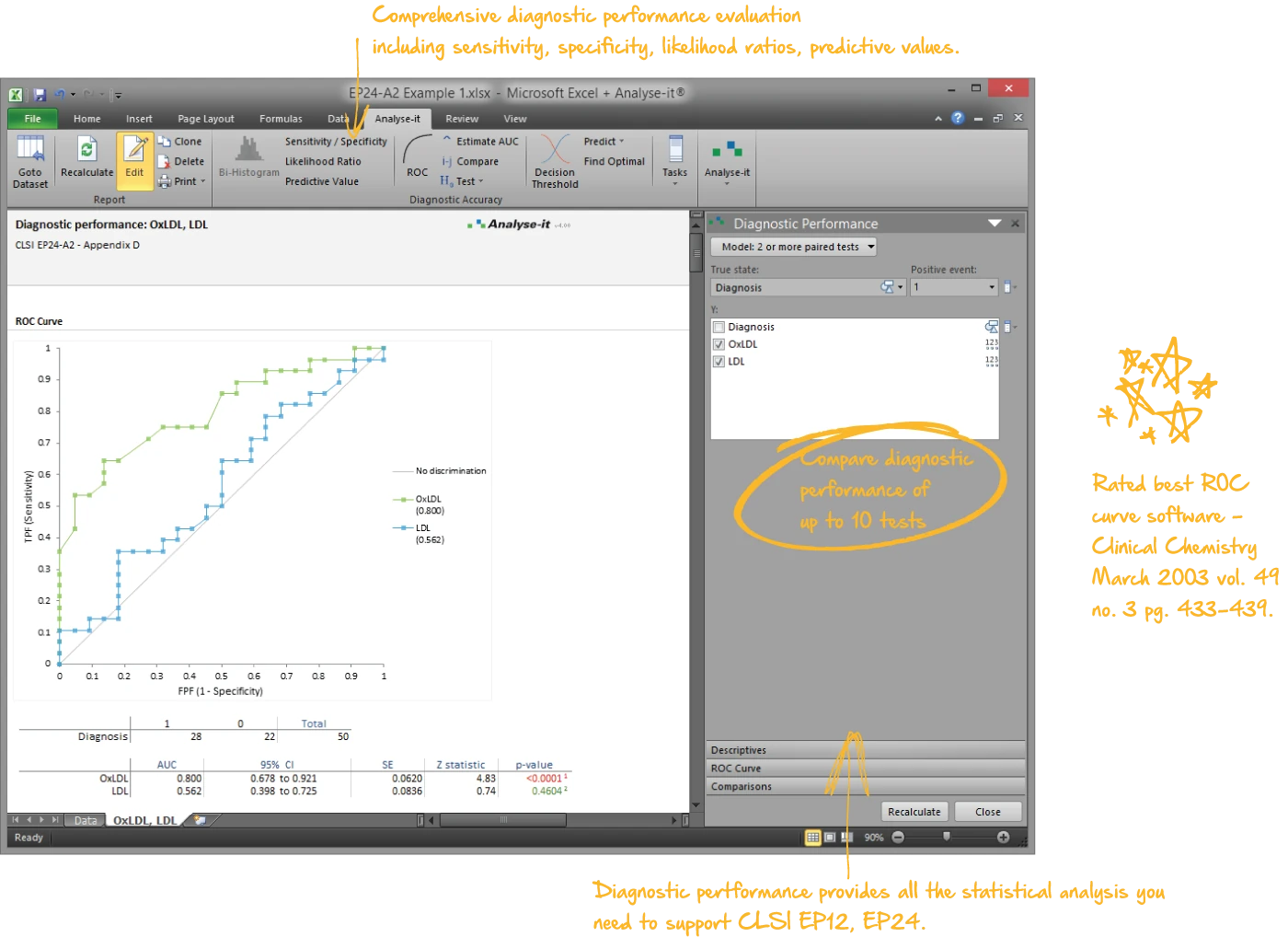
Evaluate diagnostic test accuracy
Establish and compare the ability of a diagnostic test to discriminate between outcomes. Explore how the test differentiates between positive and negative cases and determine optimum decision thresholds factoring in clinical costs of misdiagnosis.
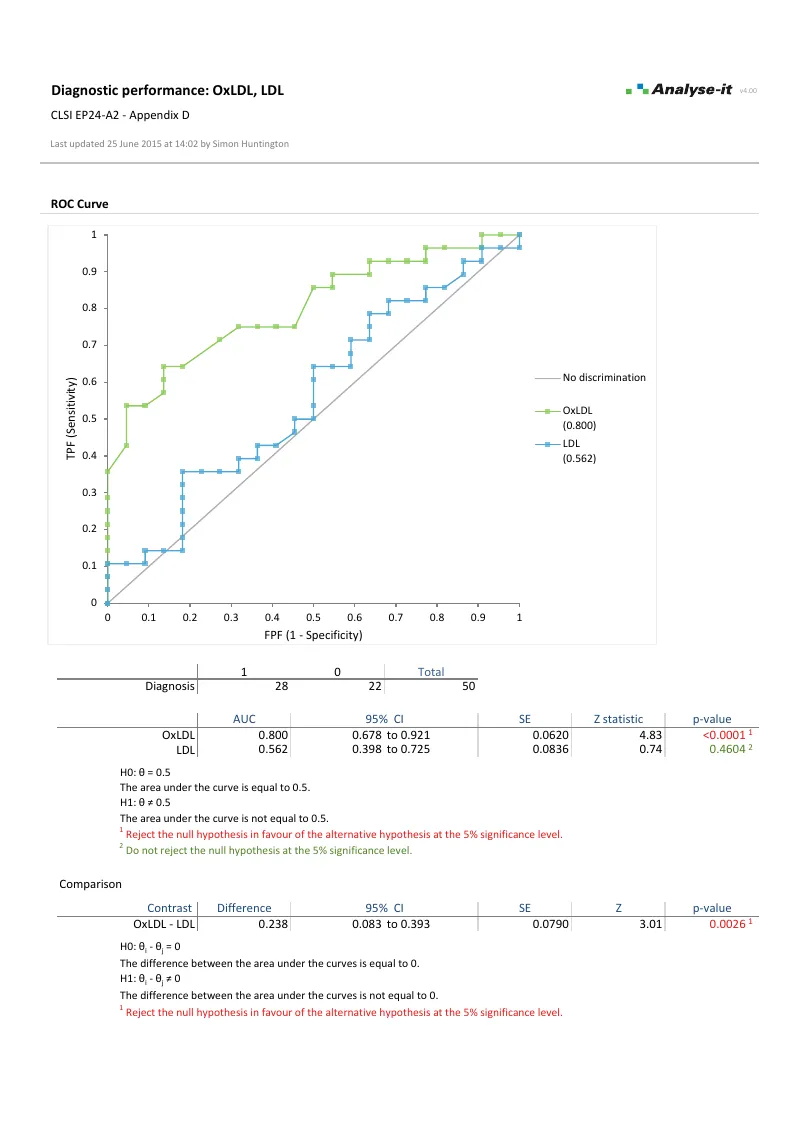
- Empirical and binormal ROC curves with DeLong AUC and confidence intervals (EP24-A2)
- AUC comparison (DeLong) for up to 10 paired or independent tests — with equality, equivalence, and non-inferiority options
- Sensitivity, specificity, likelihood ratios, predictive values, diagnostic odds ratio
- Optimal threshold — Youden, closest-to-(0,1), cost-based
- Decision plots of sensitivity vs specificity, likelihood ratios, predictive values, or cost across all possible thresholds
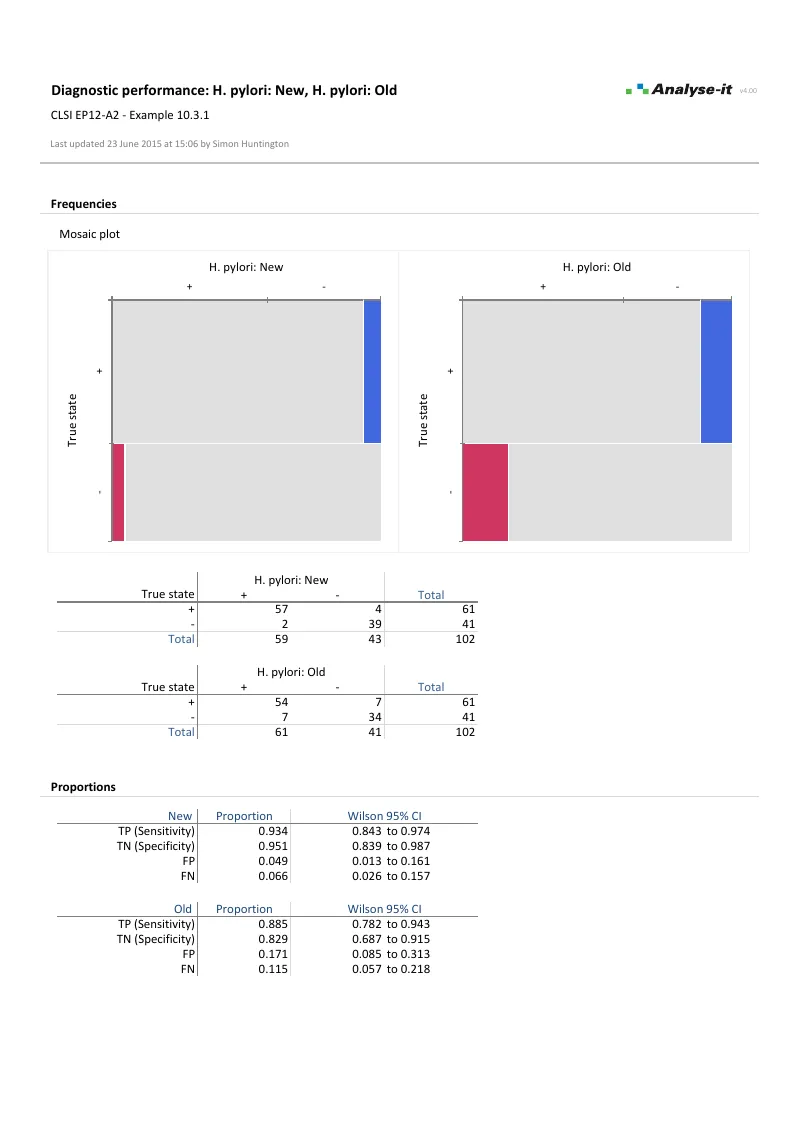
- Qualitative test evaluation — proportion in positive/negative agreement, kappa and weighted kappa (EP12-A2)
Clinical Immunology FAMH
Includes the full Standard edition
All features from the Standard edition are included — ANOVA, PCA, regression, correlation, distribution analysis, hypothesis tests, and more.
So if, or when, a validation study raises questions — an unexpected distribution, a suspect outlier, a relationship that needs further examination — the full general statistics toolkit is right there in the same workbook.
Software you can trust
Support for 11 CLSI protocols
Analyse-it has led the field in method validation statistics software for over 30 years. Many of the procedures now codified in CLSI guidelines — including Passing-Bablok regression, Weighted Deming regression, and non-parametric reference interval methods — were first made widely accessible through Analyse-it, and their adoption into formal guidelines followed.
| EP05-A3 | Evaluation of Precision of Quantitative Measurement Procedures | Precision |
| EP06-A | Evaluation of the Linearity of Quantitative Measurement Procedures | Linearity |
| EP09-A3 | Measurement Procedure Comparison and Bias Estimation Using Patient Samples | Method comparison |
| EP10-A3-AMD | Preliminary Evaluation of Quantitative Clinical Laboratory Measurement Procedures | MSA |
| EP12-A2 | User Protocol for Evaluation of Qualitative Test Performance | Diagnostic performance |
| EP14-A3 | Evaluation of Commutability of Processed Samples | Method comparison |
| EP15-A3 | User Verification of Precision and Estimation of Bias | MSA |
| EP17-A2 | Evaluation of Detection Capability for Clinical Laboratory Measurement Procedures | Detection capability |
| EP21-A | Estimation of Total Analytical Error for Clinical Laboratory Methods | Method comparison |
| EP24-A2 | Assessment of the Diagnostic Accuracy of Laboratory Tests Using Receiver Operating Characteristic Curves | Diagnostic performance |
| EP28-A3C | Defining, Establishing, and Verifying Reference Intervals in the Clinical Laboratory | Reference intervals |
Example analyses
Download CLSI example datasets, open them in the trial, and see exactly what the output looks like.
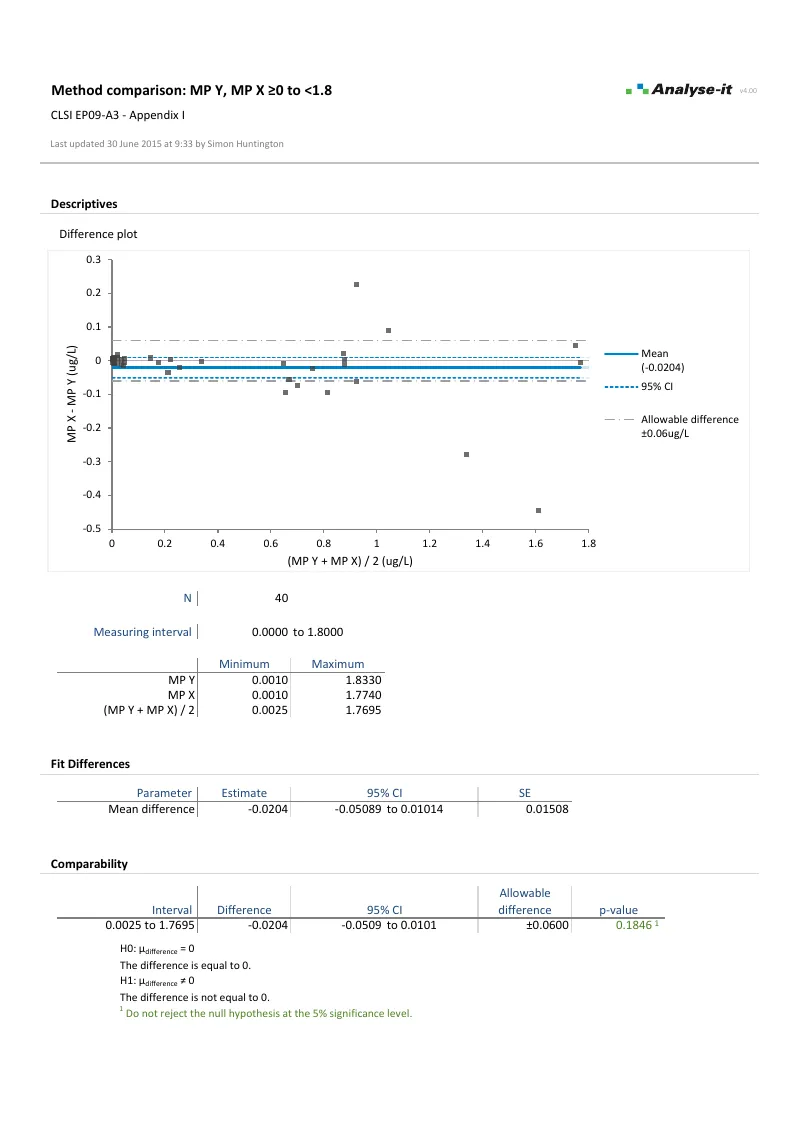 EP09-A3 — Appendix I
EP09-A3 — Appendix IBland-Altman with partitioned measuring range. 0–1.8 µg/L (allowable difference ±0.06 µg/L) and 1.8–100 µg/L (allowable difference ±6%). Mean difference with 95% CI and equality hypothesis test for each interval.
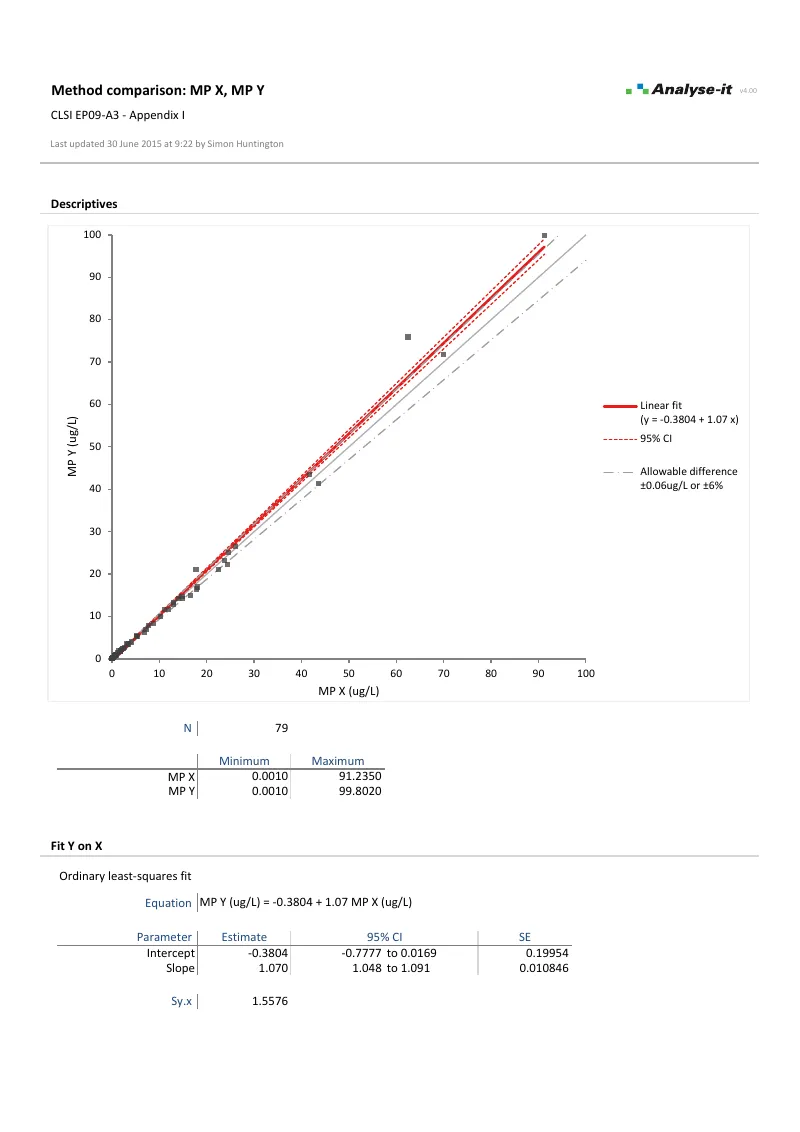 EP09-A3 — Appendix I
EP09-A3 — Appendix IAll five method comparison fits. OLS, WLS, Deming, Weighted Deming, and Passing-Bablok on 79 observations. Bias at medical decision point 5 µg/L with CI and hypothesis test for each fit.
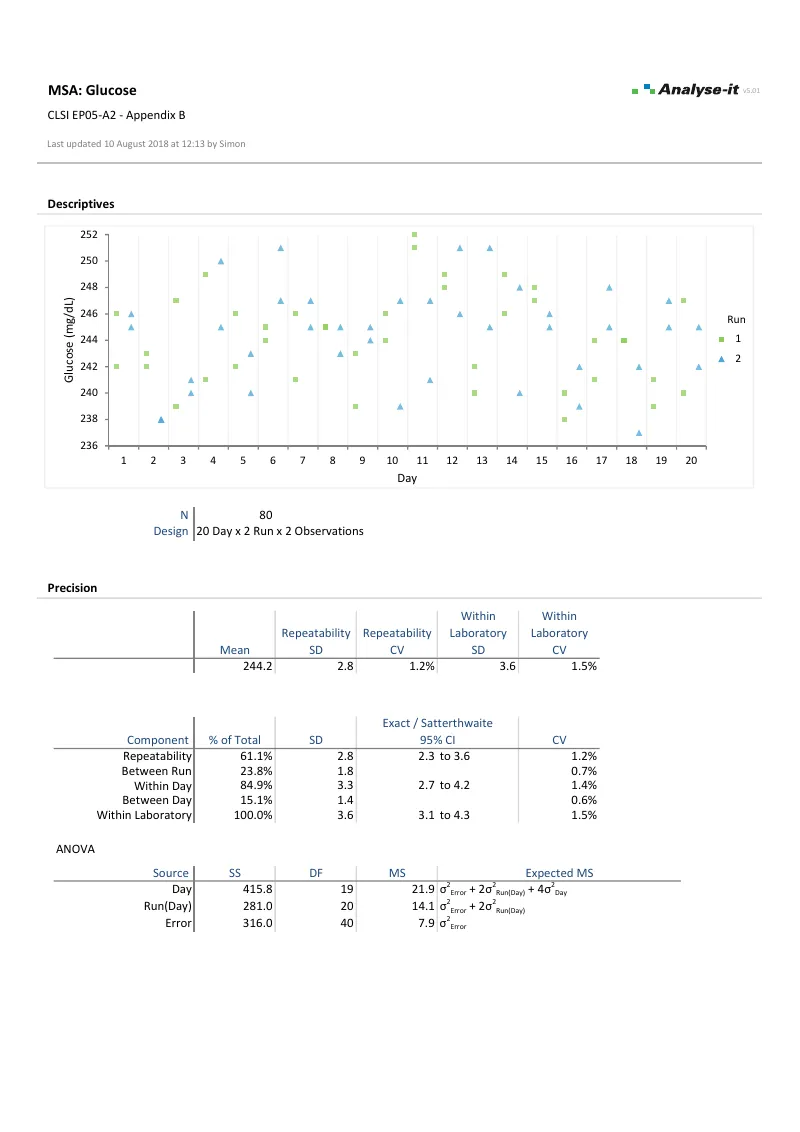 EP05-A3 — Appendix B
EP05-A3 — Appendix BGlucose precision study, single site. 20 days × 2 runs × 2 observations. Repeatability and between-run/between-day variance components, ANOVA table, and variability of measurements plot.
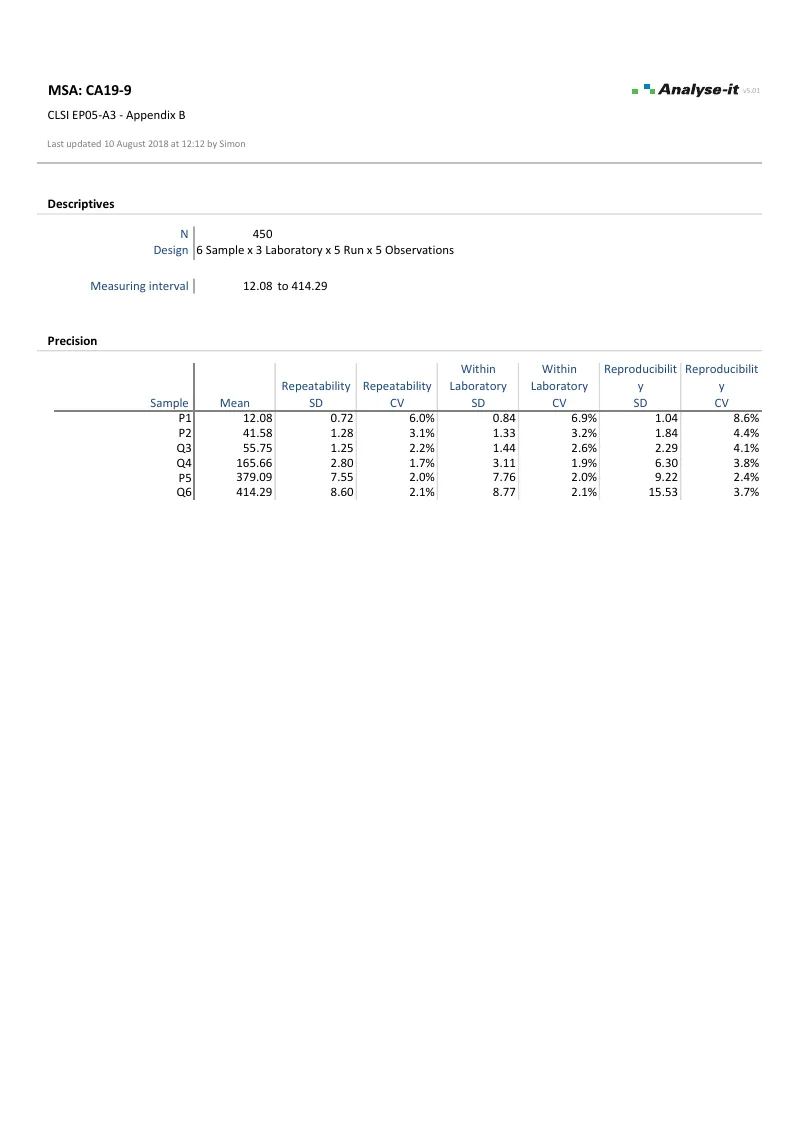 EP05-A3 — Appendix B
EP05-A3 — Appendix BCA19-9 precision study, multi-site. 6 samples × 3 laboratories × 5 runs × 5 observations. Repeatability, within-laboratory, and reproducibility components. Precision profile with 3-parameter variance function fit.
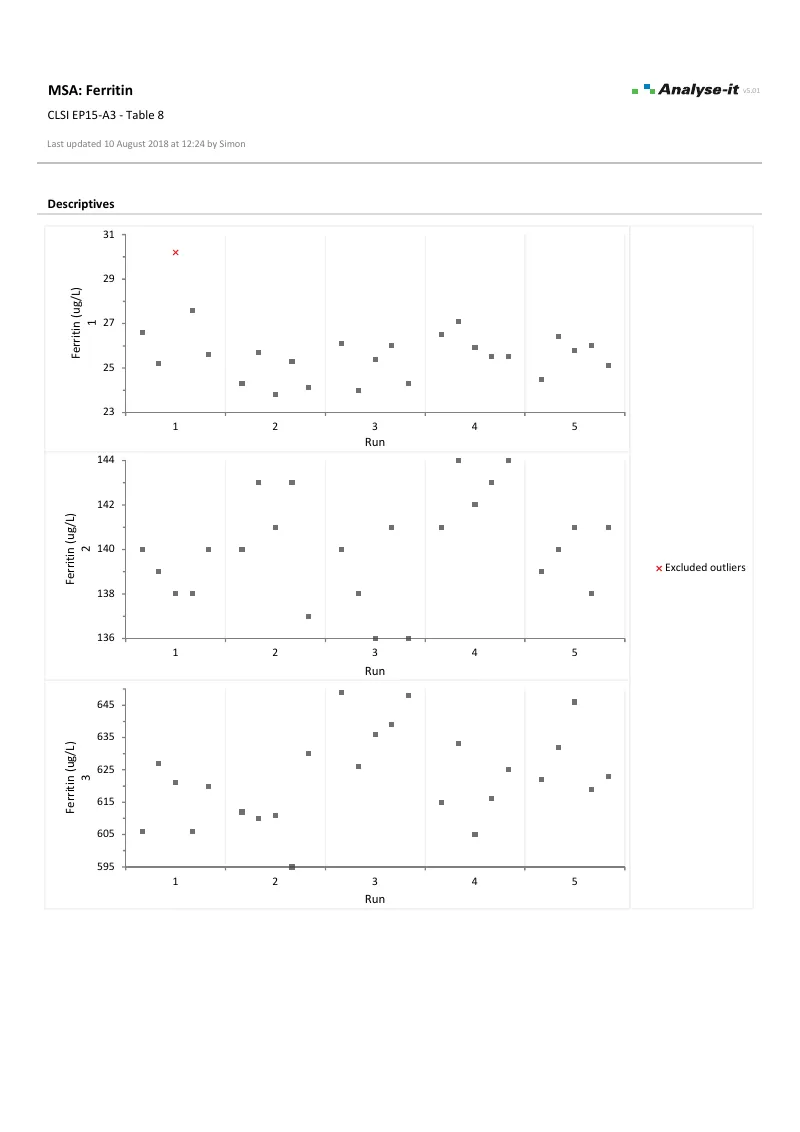 EP15-A3 — Table 8
EP15-A3 — Table 8Ferritin precision and trueness verification. 3 samples × 5 runs × 5 observations. Within-run and total precision with χ² tests against manufacturer’s claimed imprecision. Generalised ESD outlier detection.
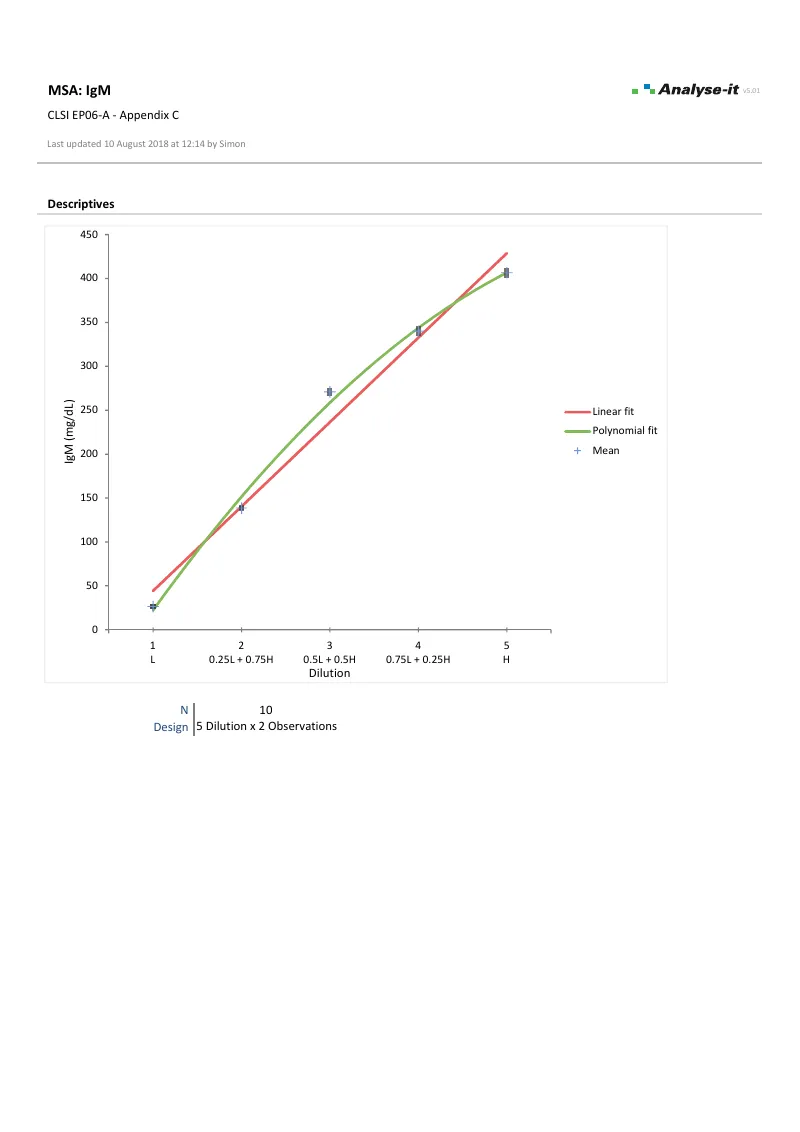 EP06-A — Appendix C
EP06-A — Appendix CIgM linearity evaluation. 5 dilutions × 2 observations. Linear and 2nd order polynomial fits. Difference plot with ±5% allowable nonlinearity band. Dilution 4 meets the requirement; dilutions 1, 2, 3, and 5 do not.
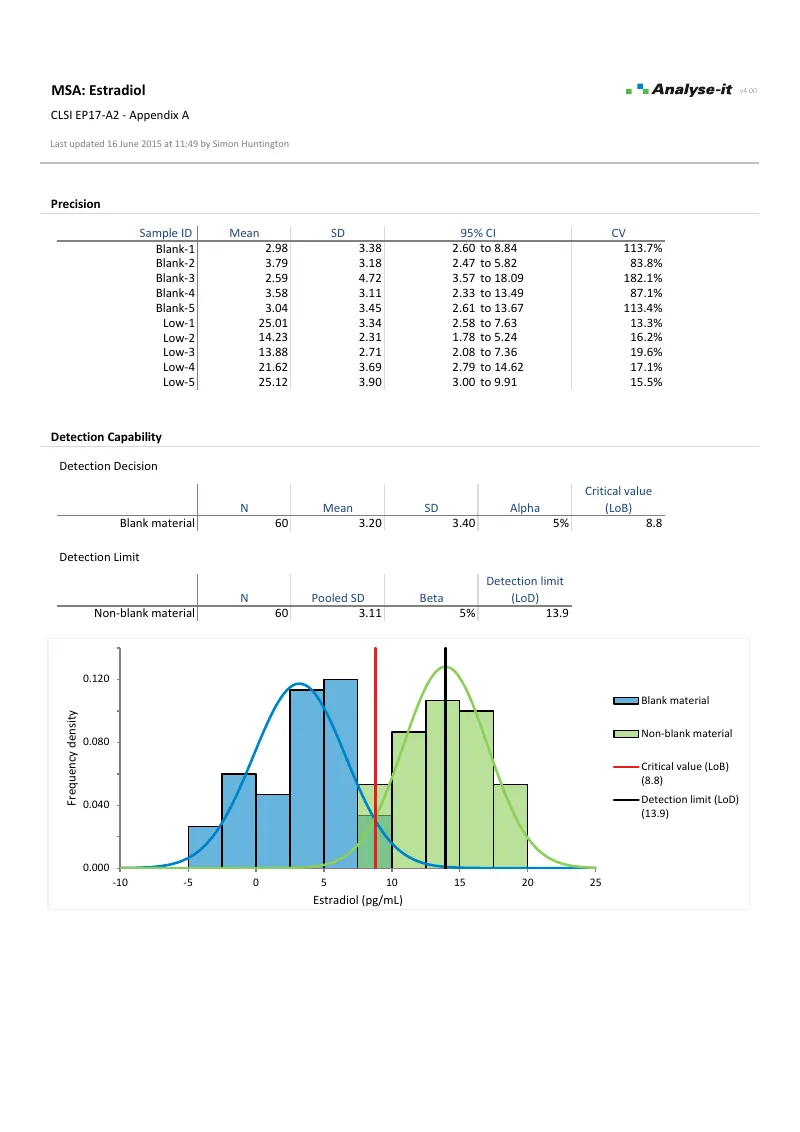 EP17-A2 — Appendix A
EP17-A2 — Appendix AEstradiol detection capability. Parametric LoB from 5 blank samples and LoD from pooled SD of 5 low-level samples. Frequency density histogram with LoB and LoD lines.
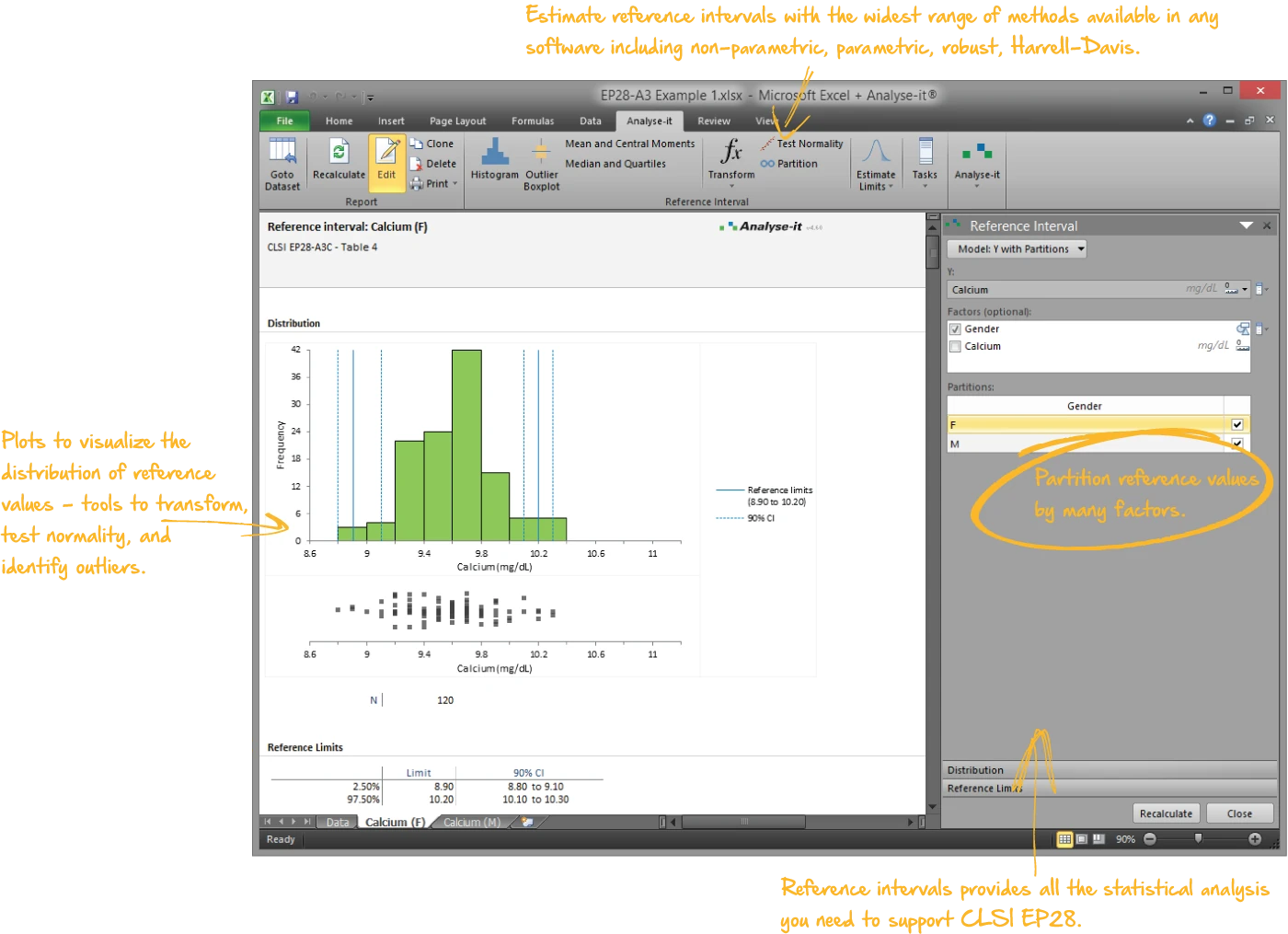
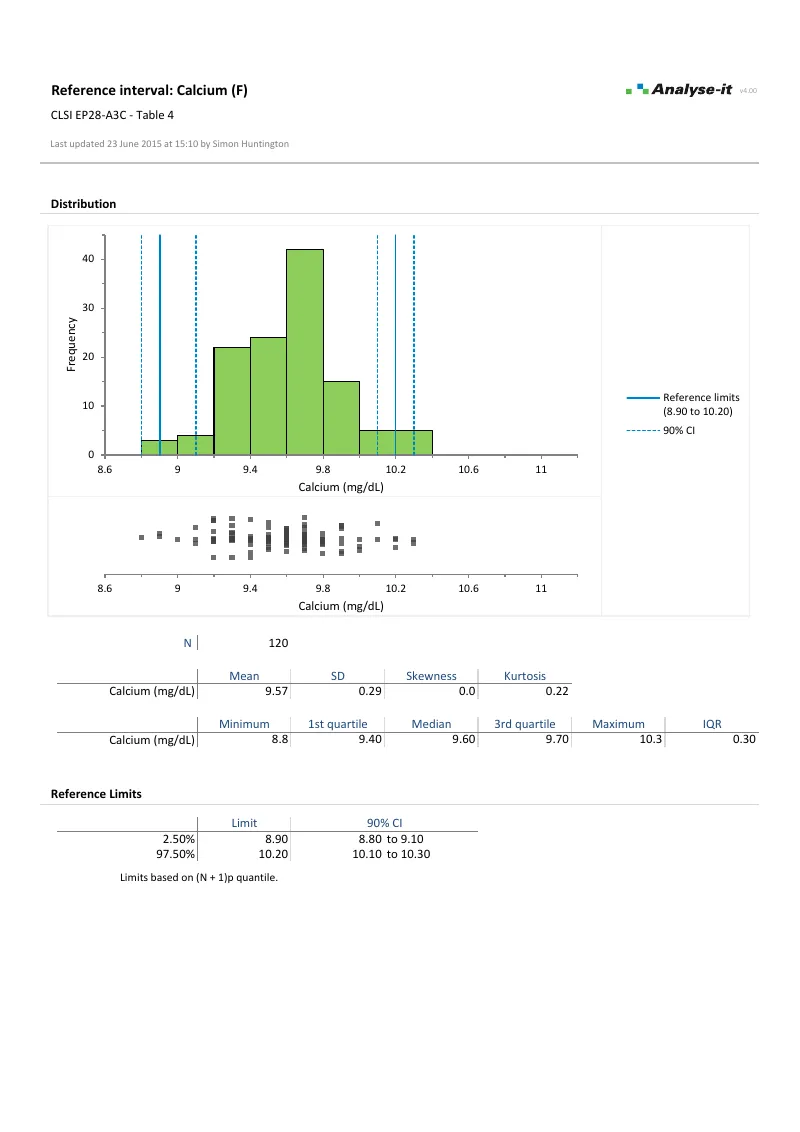 EP28-A3C — Table 4
EP28-A3C — Table 4Calcium reference intervals. Partitioned by sex. Non-parametric (N+1)p quantile method, 120 observations per group. Histogram with reference limits and 90% CIs.
Technical details
Method comparison
CLSI protocols
- EP09-A3: Measurement Procedure Comparison and Bias Estimation Using Patient Samples
- EP14-A3: Evaluation of Commutability of Processed Samples
- EP21-A: Estimation of Total Analytical Error for Clinical Laboratory Methods
Quantitative methods (EP09-A3)
- Singlicate, duplicate, and replicate measurements
- Reduce measuring interval to linear range, or partition into multiple intervals with different relationships new in v4.00
- Ordinary and Weighted linear regression average bias with confidence intervals
- Deming and Weighted Deming regression average bias with jackknife confidence intervals
- Passing-Bablok regression (1983 and 1988) average bias with Passing-Bablok or bootstrap confidence intervals
- Bland-Altman limits of agreement with mean, median, and linear fit bias new in v3.75
- Predict bias with confidence intervals at important decision levels
- Test equality (no difference) or equivalence (difference within allowable difference) at decision levels
- Compare commutability of samples with prediction intervals new in v4.90
- Precision (SD or CV) and precision plots for each method
- Pearson r correlation coefficient
Total analytical error (EP21-A)
- Combine bias from method comparison with imprecision of test method
- Assess against allowable total error
- Allowable error as absolute, percentage, or combination
Qualitative methods
- Proportion in positive/negative agreement (Clopper-Pearson exact, Wilson score CIs)
- Kappa and Weighted Kappa (Wald Z CI)
- Kappa test for agreement
Plots
- Scatter plot with average bias, confidence bands, identity line, and allowable difference band
- Scatter plot with allowable error bands
- Vary colour of points by a factor
- Difference / relative difference / ratio plot against X or mean of methods with allowable difference band and histogram
- Mountain plot with allowable difference band new in v3.71
- Residual plot (raw and standardised) with histogram
- CUSUM linearity plot and Kolmogorov-Smirnov linearity test
Measurement systems analysis
CLSI protocols
- EP05-A3: Evaluation of Precision of Quantitative Measurement Procedures
- EP06-A: Evaluation of the Linearity of Quantitative Measurement Procedures
- EP10-A3-AMD: Preliminary Evaluation of Quantitative Clinical Laboratory Measurement Procedures
- EP15-A3: User Verification of Precision and Estimation of Bias
- EP17-A2: Evaluation of Detection Capability for Clinical Laboratory Measurement Procedures
Experiment design
- Flexible balanced and unbalanced experiment design: up to 3 random nested factors new in v4.60 and 1 fixed factor
- Generalised ESD outlier identification new in v4.95
Precision (EP05-A3)
- Precision as variance, SD, or CV% with exact, Satterthwaite, and MLS confidence intervals
- Abbreviated reproducibility/repeatability, and detailed intermediate precision components
- ANOVA table
- χ² test against precision claim
- Precision profile of SD or CV
- Variance function fit: constant variance, constant CV, mixed constant/proportional, Sadler 3-parameter power, and 3-parameter alternative power new in v4.00
- 4-parameter variance function with turning point new in v4.91
Bias / trueness (EP15-A3, EP10-A3-AMD)
- Bias with confidence interval new in v4.00
- Test equality (no bias) or equivalence (bias within allowable bias)
Linearity (EP06-A)
- Linear, polynomial (up to 5th order), forward stepwise polynomial, and best polynomial regression
- Weighted models for non-constant SD across the measuring interval new in v5.00
- Adjust measuring interval to find linear range new in v5.00
- Difference between linear and nonlinear fit with Hsieh-Liu confidence intervals
- Test equality or equivalence against allowable nonlinearity
- Emancipator-Kroll linearity
Detection capability (EP17-A2) new in v4.00
- LoB: parametric (SD of blank material), non-parametric (quantile of blank material), or from precision profile variance function
- LoD: pooled SD of non-blank materials, from precision profile variance function, or probit regression
- LoQ: from precision profile variance function
Plots
- Scatter and difference plots
- Variability of measurements plot
- Precision profile with variance function
- Difference plot of bias against assigned values with allowable bias bands
- Linearity plot with polynomial fits
- Difference plot with allowable nonlinearity band
- Frequency density histogram with LoB and LoD
- Probit regression curve
Reference intervals
CLSI protocols
- EP28-A3C: Defining, Establishing, and Verifying Reference Intervals in the Clinical Laboratory
Establish reference limits
- Normal (parametric) quantile
- Non-parametric quantile: (N+1)p, Np+½, (N+⅓)p+⅓
- Harrell-Davis quantile
- Bootstrap quantile
- Robust bi-weight quantile
- Confidence intervals on all reference limits
Transfer / verify
- Transfer using method comparison regression function
- Binomial test for proportion inside reference interval
Partitioning & transformations
- Partition by factor(s) new in v4.00
- Reciprocal transformation
- Log transformation
- Square and cube root
- Box-Cox new in v3.52
- Manly exponential new in v4.00
- 2-stage exponential / modulus new in v4.00
Normality testing
- Shapiro-Wilk test
- Anderson-Darling test
- Normal Q-Q plot
Plots
- Frequency distribution histogram with normal overlay and reference limits
- Tukey outlier box plot
- Normal Q-Q plot with Lilliefors confidence band
Diagnostic performance
CLSI protocols
- EP24-A2: Assessment of the Diagnostic Accuracy of Laboratory Tests Using Receiver Operating Characteristic Curves
- EP12-A2: User Protocol for Evaluation of Qualitative Test Performance
ROC analysis (EP24-A2)
- 1 test, up to 10 paired tests, or up to 10 independent tests/groups
- Empirical and binormal ROC curves
- AUC with DeLong-DeLong-Clarke-Pearson CIs
- AUC comparison (DeLong) for paired or independent tests
- Partial AUC over specified FPR range
- Sensitivity, specificity, PPV, NPV, likelihood ratios, odds ratio, Youden index
- Optimal threshold (Youden, closest-to-(0,1), cost-based)
Qualitative tests (EP12-A2)
- Sensitivity, specificity, prevalence
- Positive/negative agreement
- Kappa and weighted kappa
Plots
- ROC curve with confidence band
- Overlaid ROC curves for test comparison
- Decision plot (sensitivity/specificity vs threshold)
- Dot plot
System requirements
- Microsoft Excel 2013, 2016, 2019, 2021, 2024 and Microsoft 365 for Microsoft Windows (32- and 64-bit)
- Microsoft Windows 8, 10, 11, Server 2016, 2019, 2022
- 2 GB RAM minimum recommended
- 80 MB disk space