Bland-Altman analysis software for method comparison and agreement Bland-Altman limits of agreement with full CLSI EP09-A3 support — mean and median bias, constant and non-constant precision, replicate measurements, interval partitioning, and confidence intervals on limits.
Try free for 15 days — no credit card required

Trusted by 75,000 scientists at most of the top-10 IVD manufacturers, and at thousands of ISO 15189, ISO/IEC 17025, and CLIA-regulated laboratories worldwide for over 30 years.






Non-constant precision handled properly
When the spread of differences widens across the measuring range, standard limits of agreement are wrong. Analyse-it detects non-constant precision and calculates V-shaped limits that widen with concentration — exactly as the clinical reality requires.
Validated against CLSI EP09-A3 data
Every calculation runs in Analyse-it's own engine — no Excel formulas, no third-party functions. Results are validated against the example datasets published in the CLSI EP09-A3 guidelines before every release. Defensible in a 510(k), CE-IVD technical file, CAP inspection, or ISO 15189 audit.
Data stays in your facility
Analyse-it runs entirely within Microsoft Excel on your PC. No cloud processing, no data transmission — your pre-submission and patient-adjacent data stays within your facility under your data governance controls.
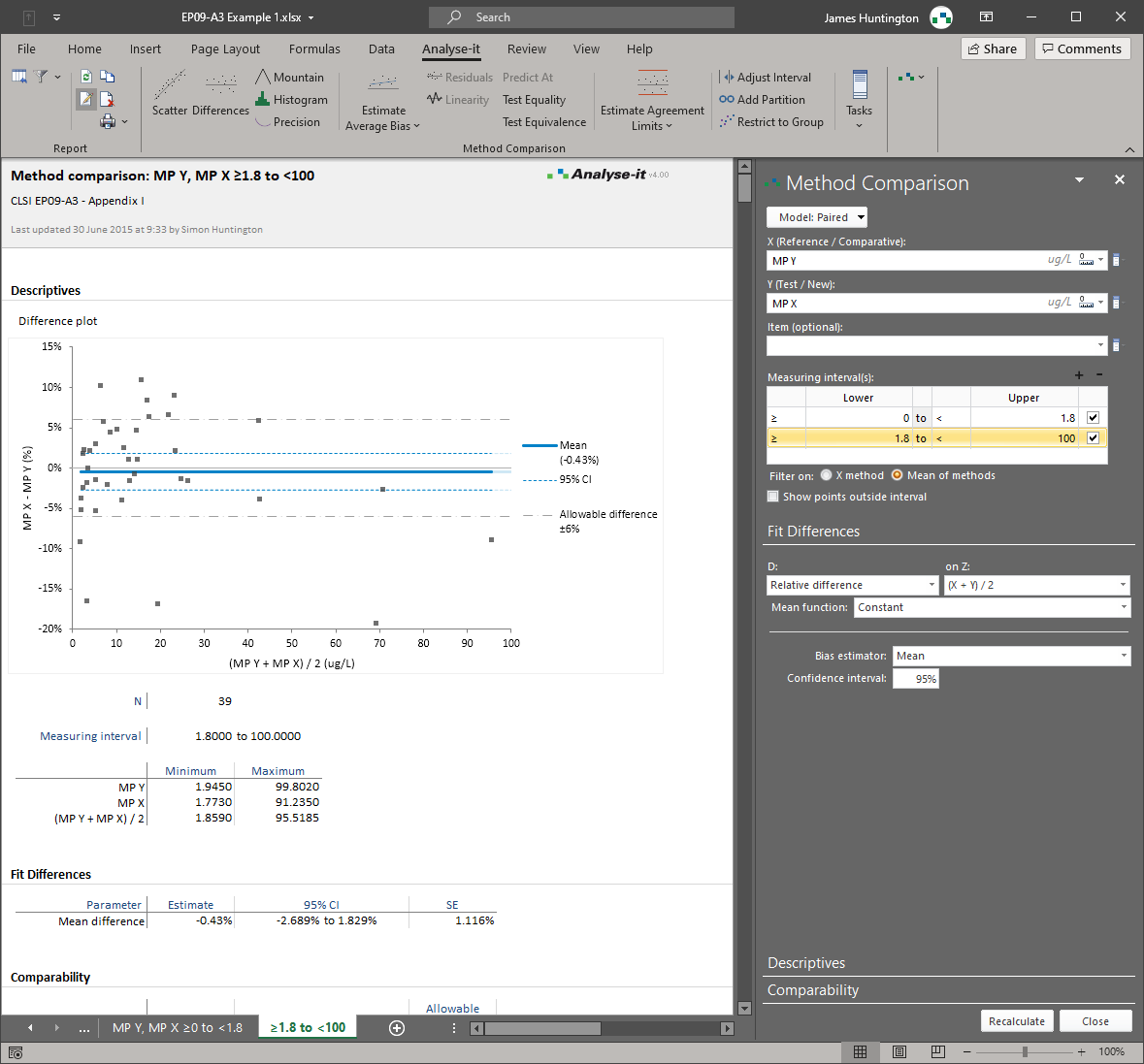
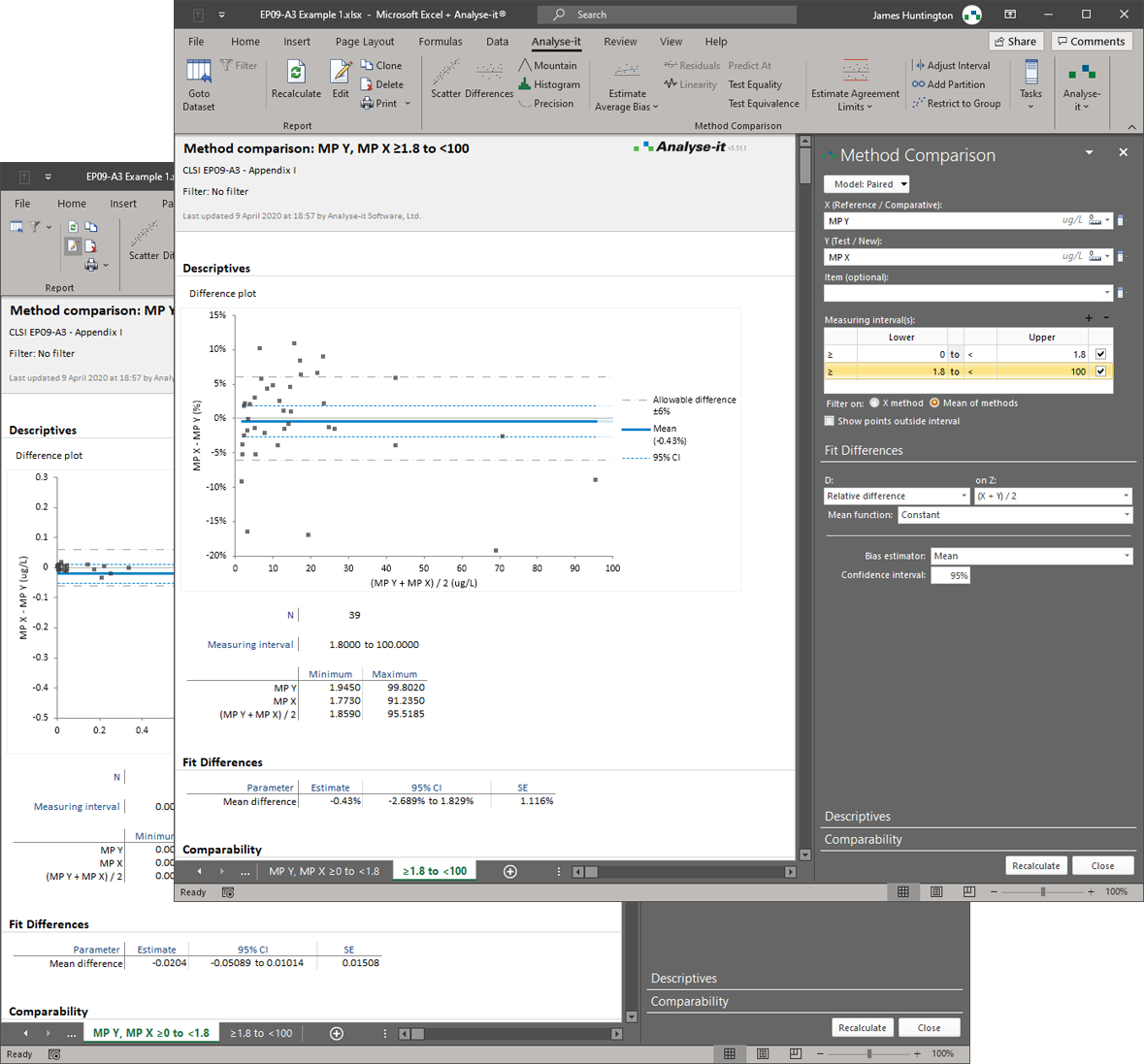
Most Bland-Altman implementations produce a difference plot with horizontal limits of agreement and stop there. That covers the simplest case — constant bias, constant precision, no replicates. In practice, clinical method comparisons are rarely that clean. Precision often varies across the measuring range, producing a fan-shaped pattern of differences that horizontal limits fail to capture. Replicate measurements require correct within-subject variance estimation rather than naive averaging. And when precision behaviour differs between low and high concentrations, the analysis needs to be partitioned into intervals. These are the cases where basic implementations mislead, and where CLSI EP09-A3 sets the standard for what a proper analysis requires.
Analyse-it covers the full Bland-Altman protocol specified in EP09-A3. Mean bias and median bias with confidence intervals. Limits of agreement for constant precision (horizontal) and non-constant precision (V-shaped limits that widen proportionally). A linear trend fit to the differences to detect proportional bias. Replicate measurements with correct within-subject variance estimation and appropriately adjusted limits. Interval partitioning by sex, age, instrument, or any other factor when a single analysis across the full range is inappropriate. Confidence intervals on the limits of agreement themselves, so you can assess the uncertainty in the agreement boundary — not just the agreement estimate.
Bland-Altman analysis sits alongside Passing-Bablok, Deming, Weighted Deming, OLS, and Weighted OLS regression in the same method comparison analysis. Run the regression to quantify bias, then switch to Bland-Altman to see how the differences are distributed in practice — whether the disagreement is uniform or concentrated in a specific part of the range, whether there are outliers that deserve investigation, and whether the limits of agreement are clinically acceptable. All on the same data, in the same workbook, without re-entering anything.
Bland-Altman analysis in Analyse-it is used well beyond IVD validation. Clinical labs use it for analyser verification before go-live. Researchers use it for agreement studies in published work. The audience is broader than any other method comparison feature — but the requirement is the same: a complete implementation that handles the real-world cases, not just the textbook example. Scientists at Abbott, Roche, Thermo Fisher, and thousands of clinical laboratories and research institutions worldwide have relied on Analyse-it for over 30 years.
Seen in the field
What's included
-
Mean and median bias with confidence intervals
Estimate bias using the mean or median of differences. Confidence intervals on the bias estimate and on the limits of agreement, so you can assess the precision of the agreement boundary itself — not just the point estimate.
-
Limits of agreement for constant and non-constant precision
Horizontal limits when precision is constant. V-shaped limits that widen proportionally when precision varies across the measuring range. A linear trend fit to the differences to detect and quantify proportional bias.
-
Correct handling of replicate measurements
Handle singlicate, duplicate, and replicate measurements correctly. Within-subject variation is properly estimated and limits of agreement adjusted accordingly — not approximated by averaging replicates.
-
Partition by measuring interval, sex, age, or instrument
When a single analysis across the full range is inappropriate, partition the data by one or more factors and apply separate Bland-Altman analyses to each interval. Exactly as EP09-A3 specifies for non-constant precision.
-
Assess clinical impact at decision thresholds
Evaluate how the differences between methods affect interpretation at clinically important decision points. Determine whether the level of agreement is acceptable where it matters most — at the concentrations that change patient management.
-
Validated engine you can defend at inspection
Every calculation runs in Analyse-it's own engine — no Excel formulas or third-party functions. Validated against the CLSI EP09-A3 published example datasets before every release. See the development and validation process.
-
Part of the Method Validation edition
Bland-Altman analysis is included in the Method Validation edition, which covers the full CLSI method validation workflow — measurement system analysis (EP05-A3, EP06-A, EP15-A3, EP17-A2), method comparison (EP09-A3, EP21-A), reference intervals (EP28-A3C), diagnostic performance (EP24-A2, EP12-A2), and total analytical error (EP21-A). One tool for everything your laboratory or IVD submission requires.
-
We use Analyse-it frequently for our verification and pre-verification work, in accordance with CLSI guidelines for in-vitro diagnostics. It's saved time and effort compared to the hodge-podge of applications we used before, JMP, SAS, etc...Brian Noland, Ph.D.Principal Scientist, Product DevelopmentBiosite / Inverness Medical Innovations
-
We use Analyse-it for the analysis of data necessary to file 510k. We chose Analyse-it because it works in Excel, includes CLSI protocols, and, unlike EP-Evaluator, lets us analyze data directly from equipment without typing.Thomas D Harrigan, Ph.D.Technical Product ManagerAlfa Wassermann Diagnostic Technologies
-
I used Analyse-It for many product development, product troubleshooting, and technology evaluation activities... your product was the easiest to use, was accurate, and produced publication ready reports.Stanley F. Cernosek, Ph.D.Clinical Chemistry Reagent DevelopmentBeckman Coulter, Inc.
-
Analyse-it has been a tremendous help. I've published and presented at national cardiology meetings and couldn't have accomplished most of my research without it. Using Analyse-it, I even found errors or omissions in the work of our statistician!Regina S. Druz, MD, FACC, FASNCDirector, Nuclear CardiologyNorth Shore University Hospital
-
Although we only scratch the surface of Analyse-it’s capabilities, we have a very high volume of use for the statistics we need. It’s saved us time and the reports look professional.Michael SavageChemistry SupervisorBaptist Hospital East
Trusted by 75,000 scientists at most of the top-10 IVD manufacturers, and at thousands of ISO 15189, ISO/IEC 17025, and CLIA-regulated laboratories worldwide for over 30 years.
Try free for 15 days — no credit card required
See all Method Validation features
Pricing and licensing
Reference lab case study