Every statistical tool we make, in one licence Method validation with 11 CLSI protocols, diagnostic accuracy with ROC curves, survival analysis with Kaplan–Meier and Cox regression, statistical process control with Shewhart, CUSUM, and EWMA charts, plus the full general statistics toolkit — ANOVA, regression, PCA, and more.
Most organisations don’t fit neatly into a single edition. The IVD company running CLSI method validation studies also needs SPC on the manufacturing line. The clinical lab verifying a new assay also runs survival analysis for research publications. The quality team monitoring control charts also needs ANOVA and regression to investigate out-of-control events.
The Ultimate edition unlocks every feature across all four editions — Method Validation, Medical, Quality Control & Improvement, and Standard — in a single licence at US$575/yr, less than any two specialist editions bought separately. Already own a specialist edition? Upgrade for the difference in price.
Principal Scientist, Product Development
Biosite / Inverness Medical Innovations
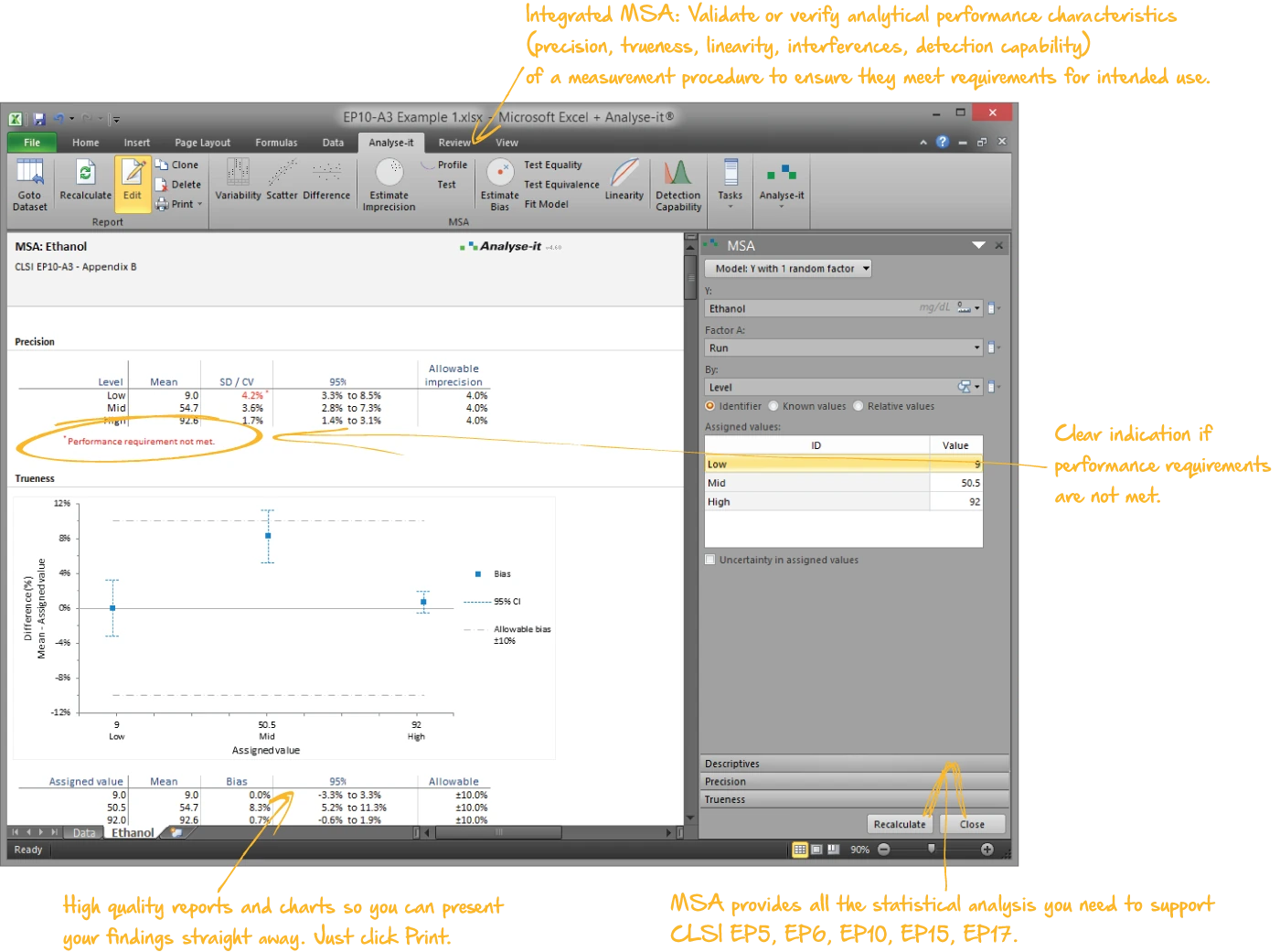
Validate and verify measurement system performance
The complete CLSI method validation toolkit — precision, linearity, detection capability, method comparison, reference intervals, and diagnostic performance — with 11 evaluation protocols built in:
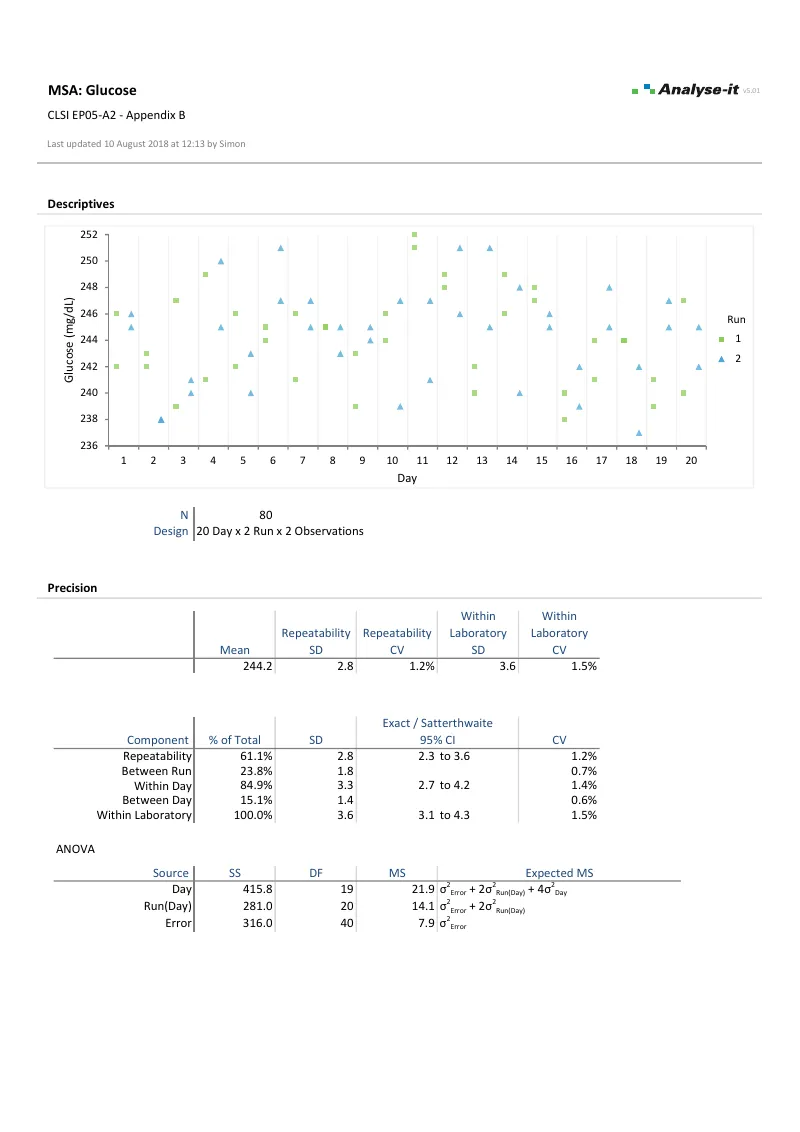
- Precision with nested variance components, precision profiles, and five variance function models (EP05-A3)
- Linearity with polynomial and weighted regression, Hsieh-Liu nonlinearity testing, and measuring interval (EP06-A)
- Detection capability — LoB, LoD, LoQ using parametric, non-parametric, probit regression, and variance function approaches (EP17-A2)
- Verification of precision and trueness against manufacturer claims (EP15-A3), interference testing (EP10-A3-AMD)
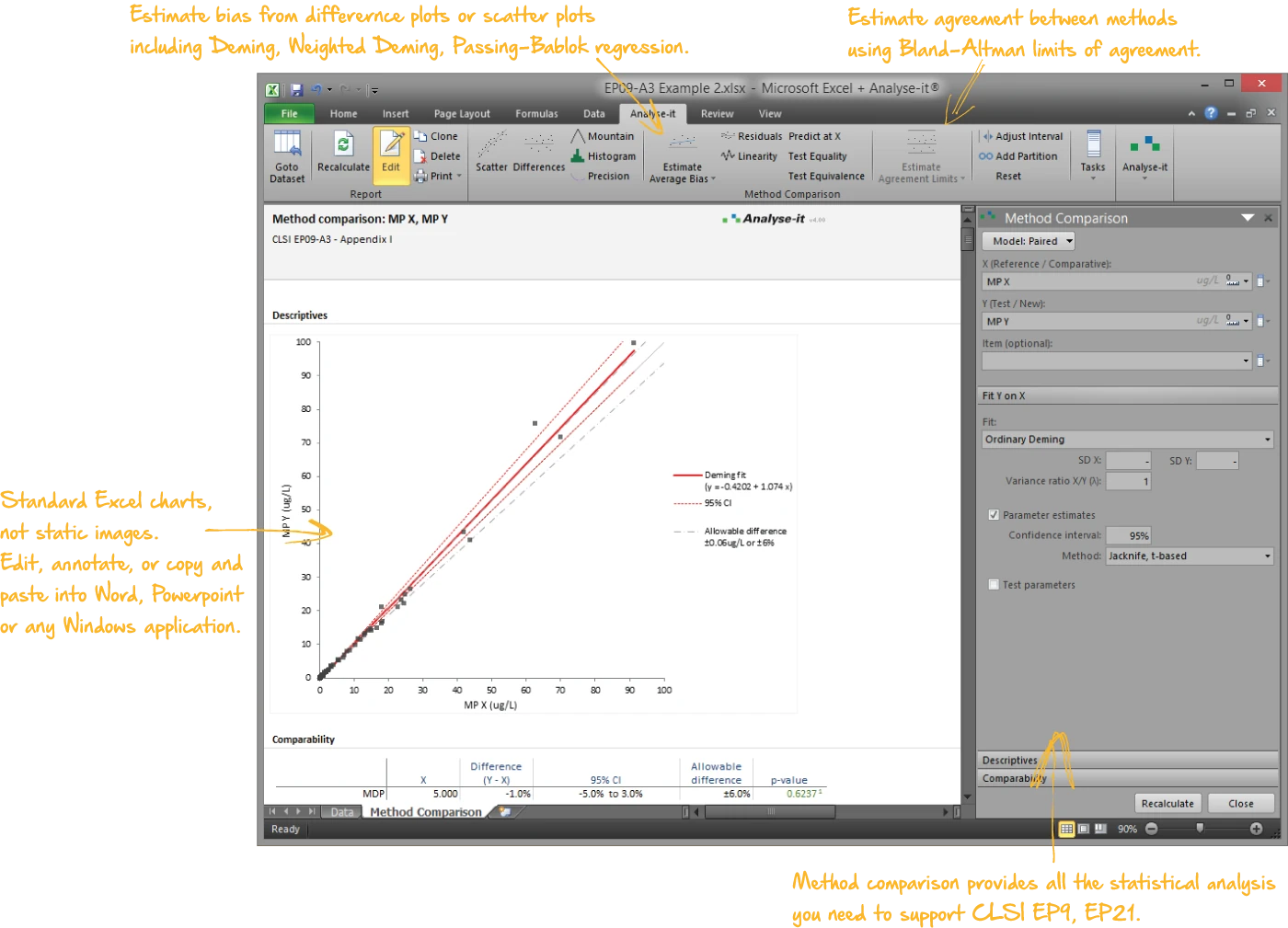
Compare methods and evaluate the impact of changes
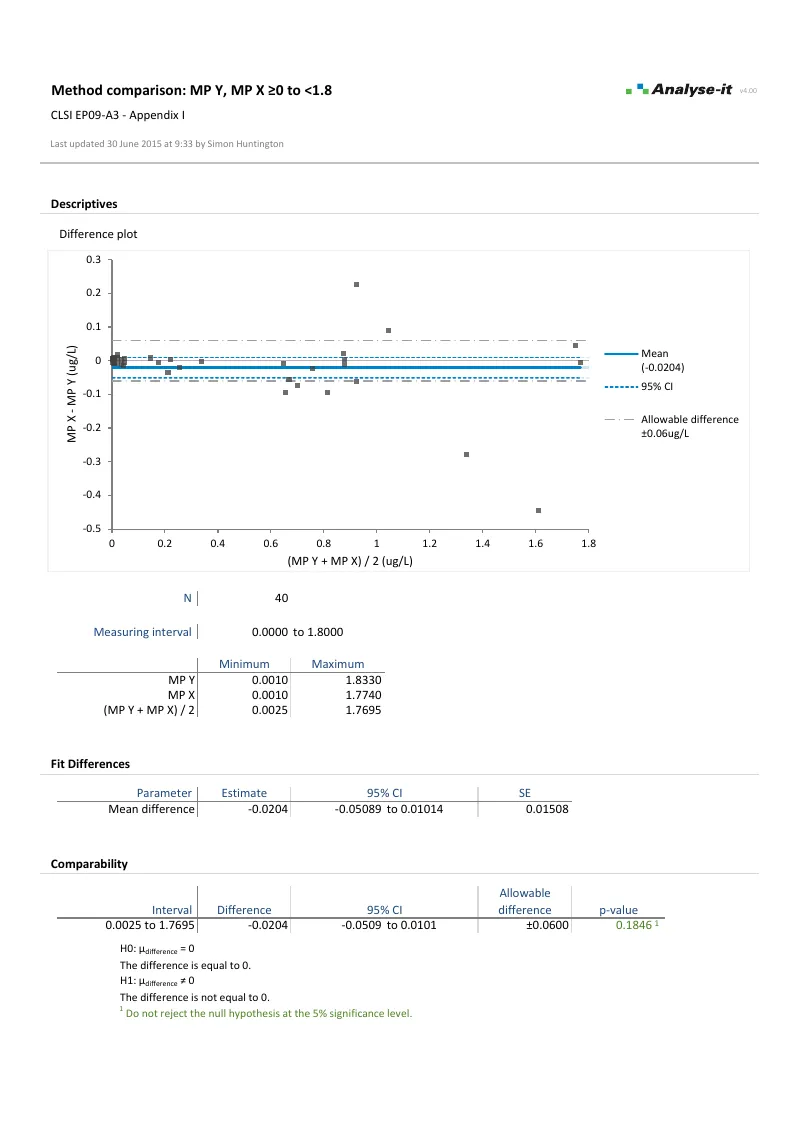
Five regression models for method comparison — including Passing-Bablok, Deming, and weighted Deming — plus Bland–Altman agreement and total analytical error:
- Passing-Bablok and Deming regression with confidence intervals on slope and intercept, medical decision point evaluation
- Bland–Altman limits of agreement — mean, median, and regression-based bias with constant and non-constant precision
- Total analytical error against allowable limits (EP21-A)
- Commutability assessment for reference materials (EP14-A3)
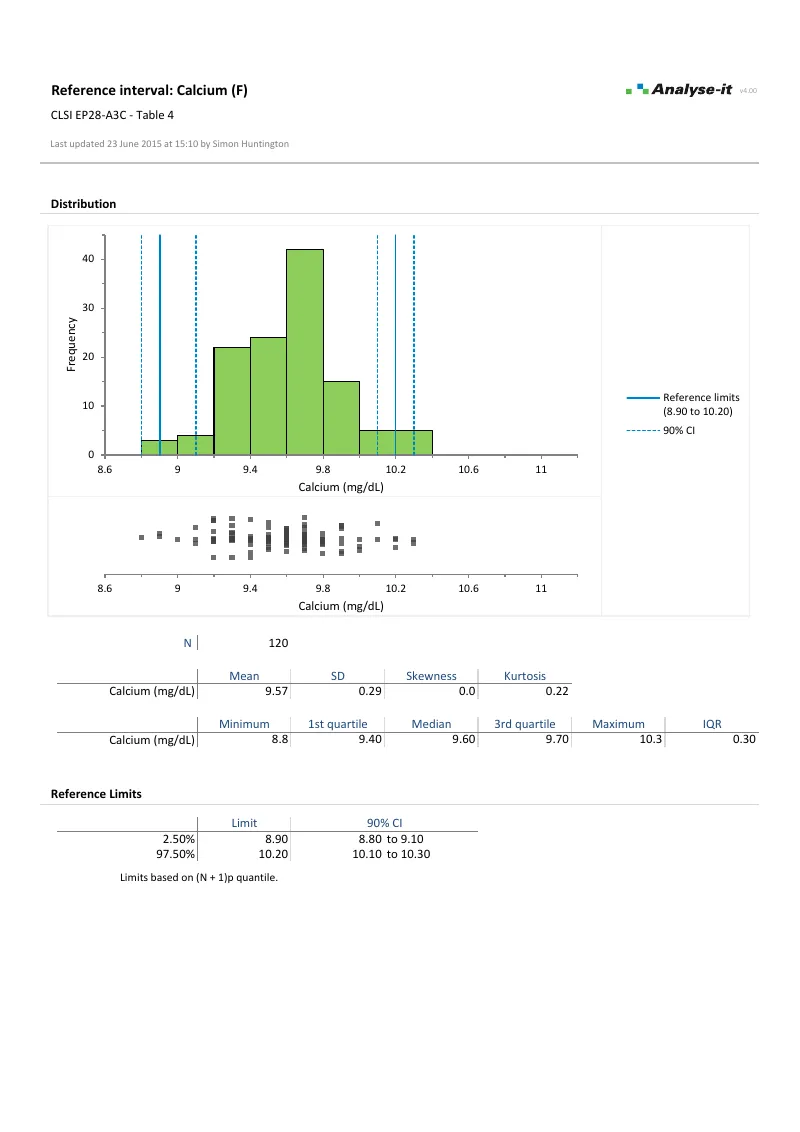
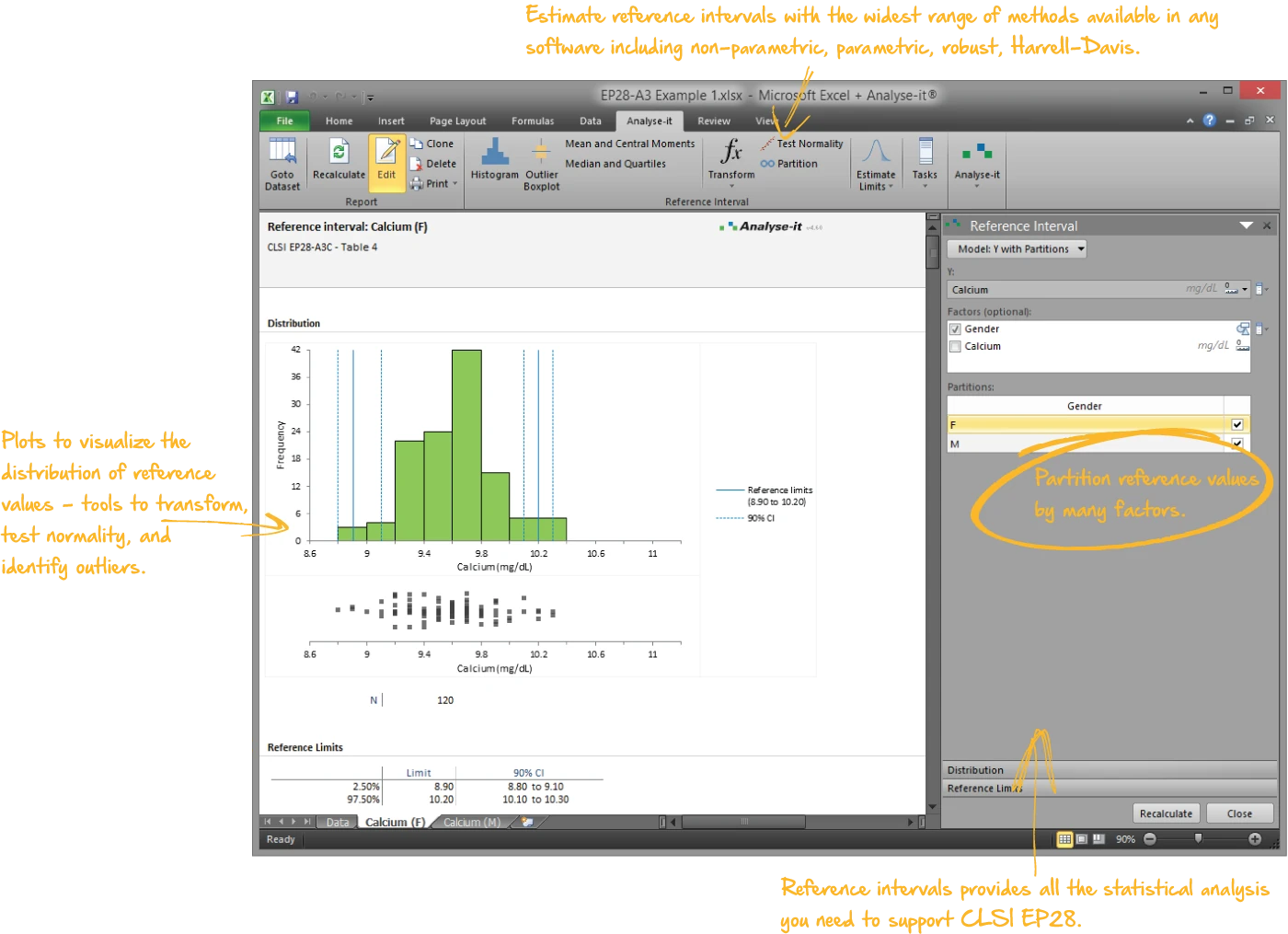
Establish reference intervals for clinical interpretation
Full range of reference interval methods — match the method to your sample size and distribution, partition where subgroups need separate ranges, and transfer or verify intervals when moving to a new measurement procedure:
- Five quantile methods — parametric, non-parametric (three computation approaches), robust bi-weight, bootstrap, and Harrell–Davis
- Partition by sex, age, ethnicity, or any combination of factors
- Full range of transformations — log, square root, cube root, Box–Cox, Manly exponential, two-stage exponential/modulus
- Transfer and verify existing intervals using regression or binomial proportion test
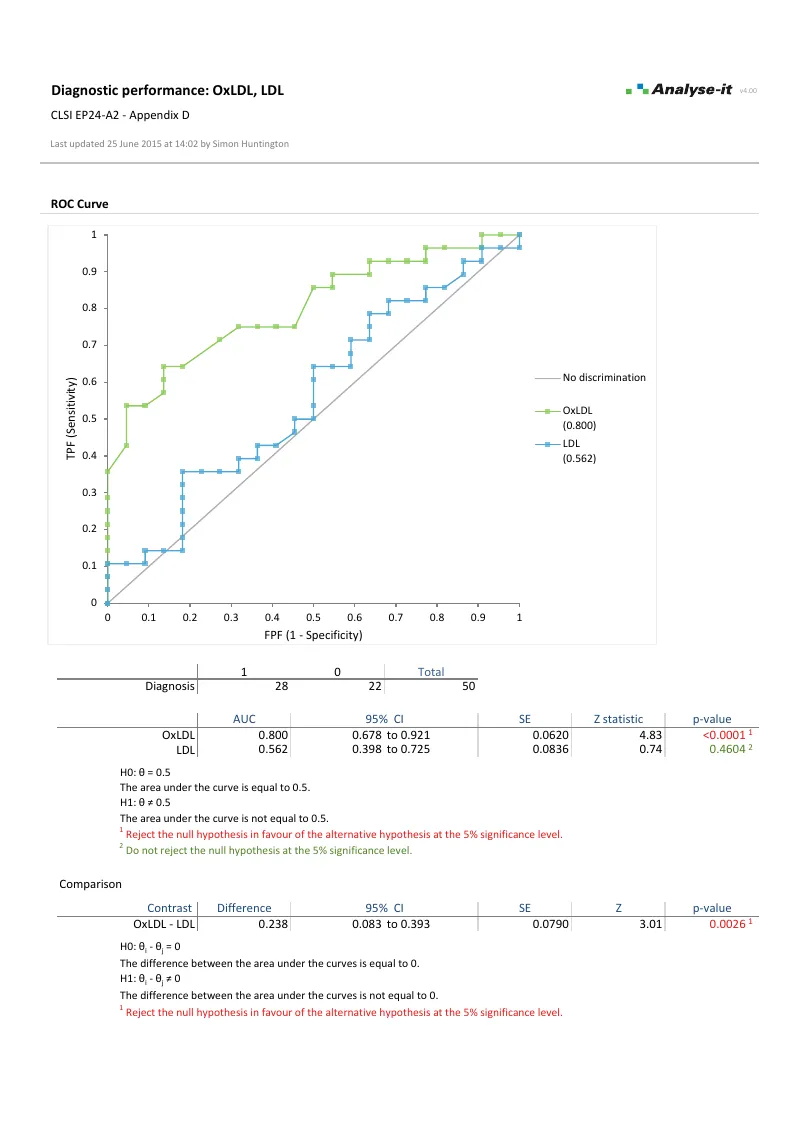
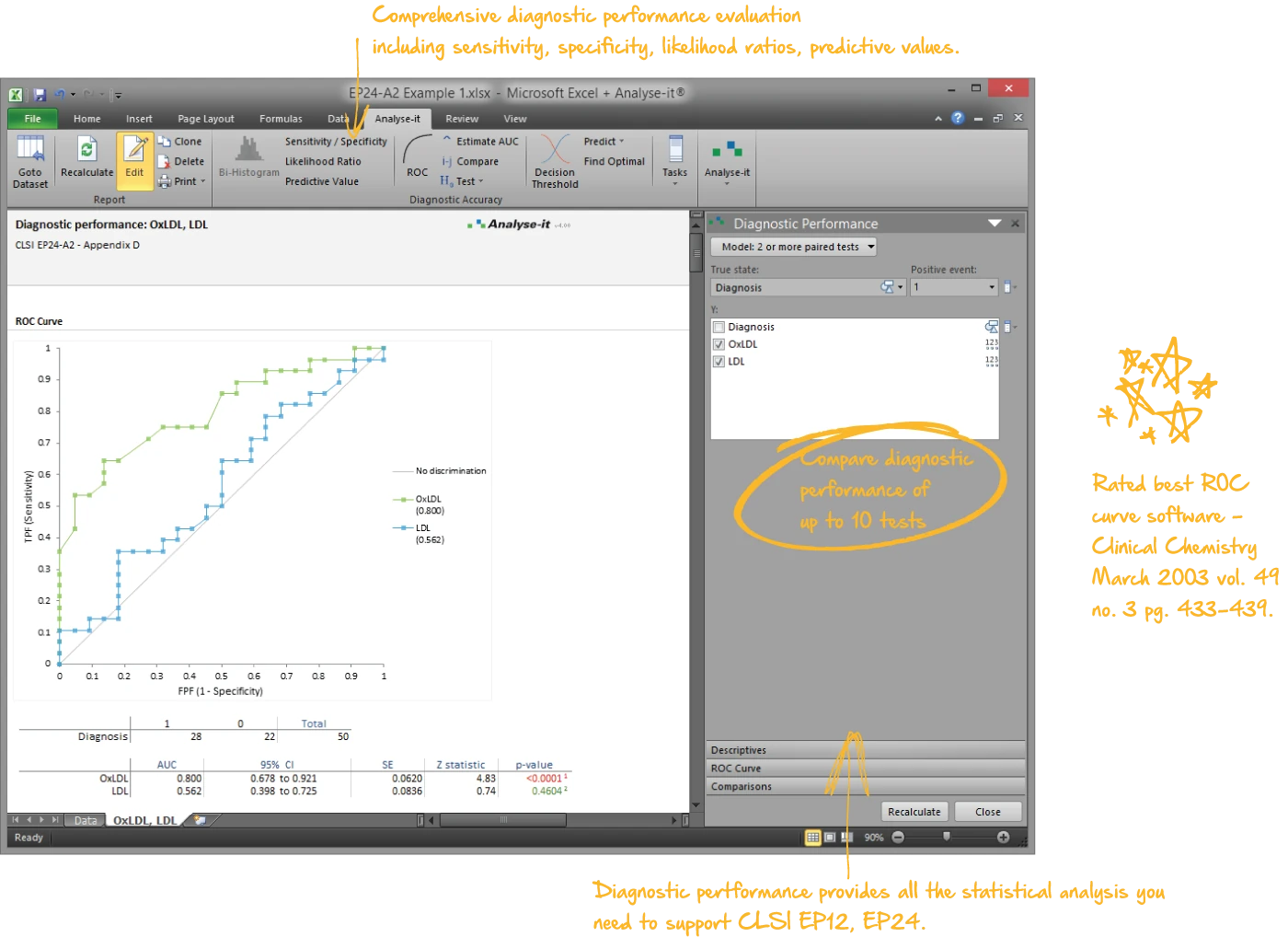
Evaluate diagnostic test accuracy
Establish and compare the ability of a diagnostic test to discriminate between patients with and without a condition — and find the decision threshold that balances sensitivity, specificity, and clinical cost:
- Empirical and binormal ROC curves with DeLong AUC and confidence intervals (EP24-A2)
- AUC comparison (DeLong) for up to 10 paired or independent tests
- Sensitivity, specificity, likelihood ratios, predictive values, diagnostic odds ratio
- Optimal threshold — Youden, closest-to-(0,1), cost-based
- Qualitative test evaluation — PPA/NPA, kappa, weighted kappa (EP12-A2)
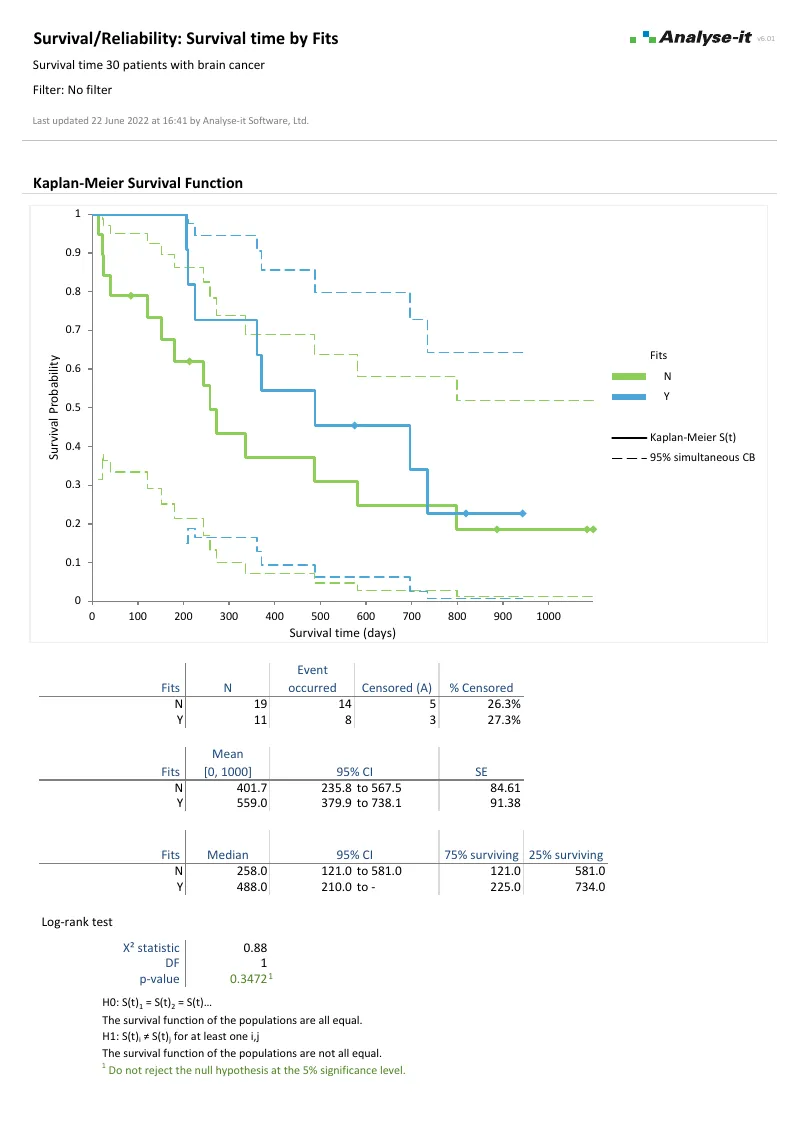
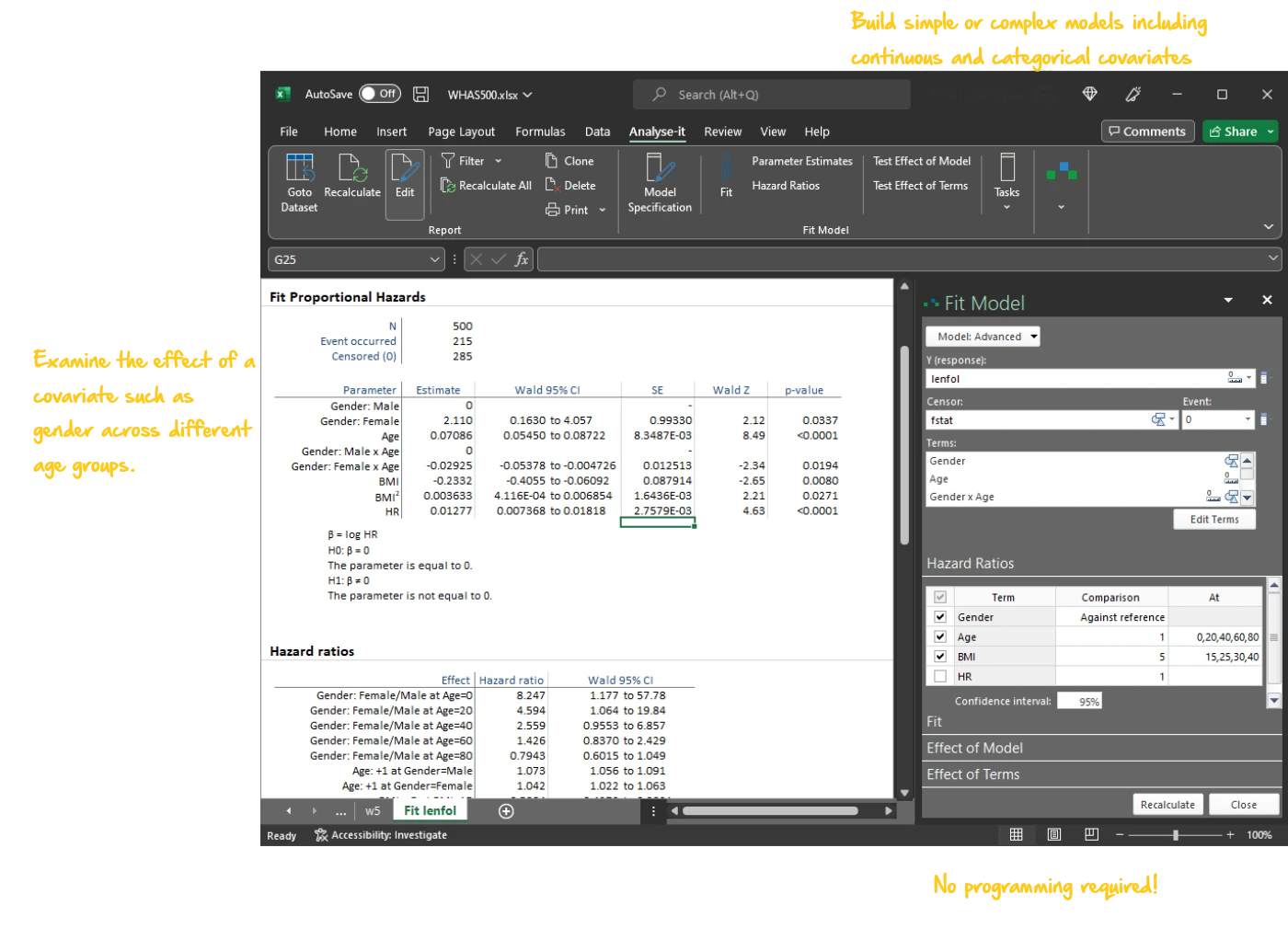
Analyse time-to-event data with survival analysis
Estimate survival functions, compare treatment groups, and model the effect of covariates on the hazard:
- Kaplan–Meier survival curve with pointwise, Nair, or Hall–Wellner confidence bands
- Median, quartile, and restricted mean survival time
- Test equality of survival functions with log-rank, Wilcoxon, Tarone–Ware, Fleming–Harrington
- Cox proportional hazards regression — hazard ratios with confidence intervals, baseline survival function
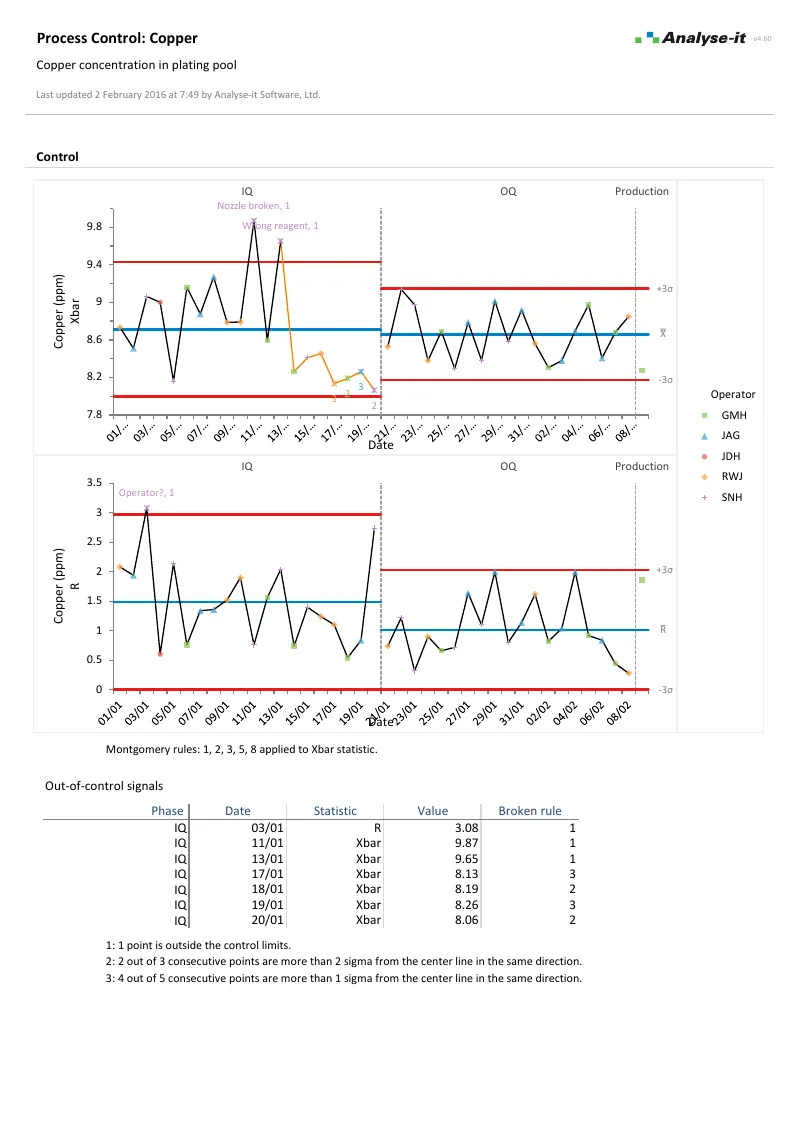
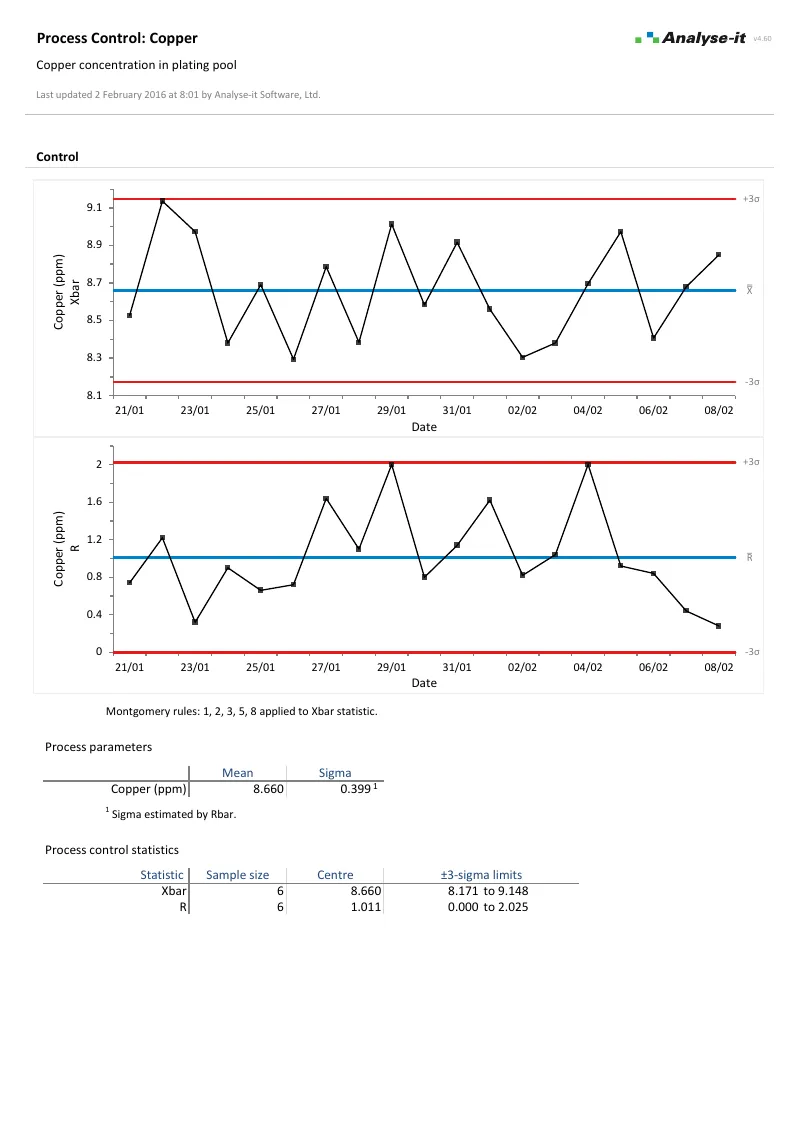
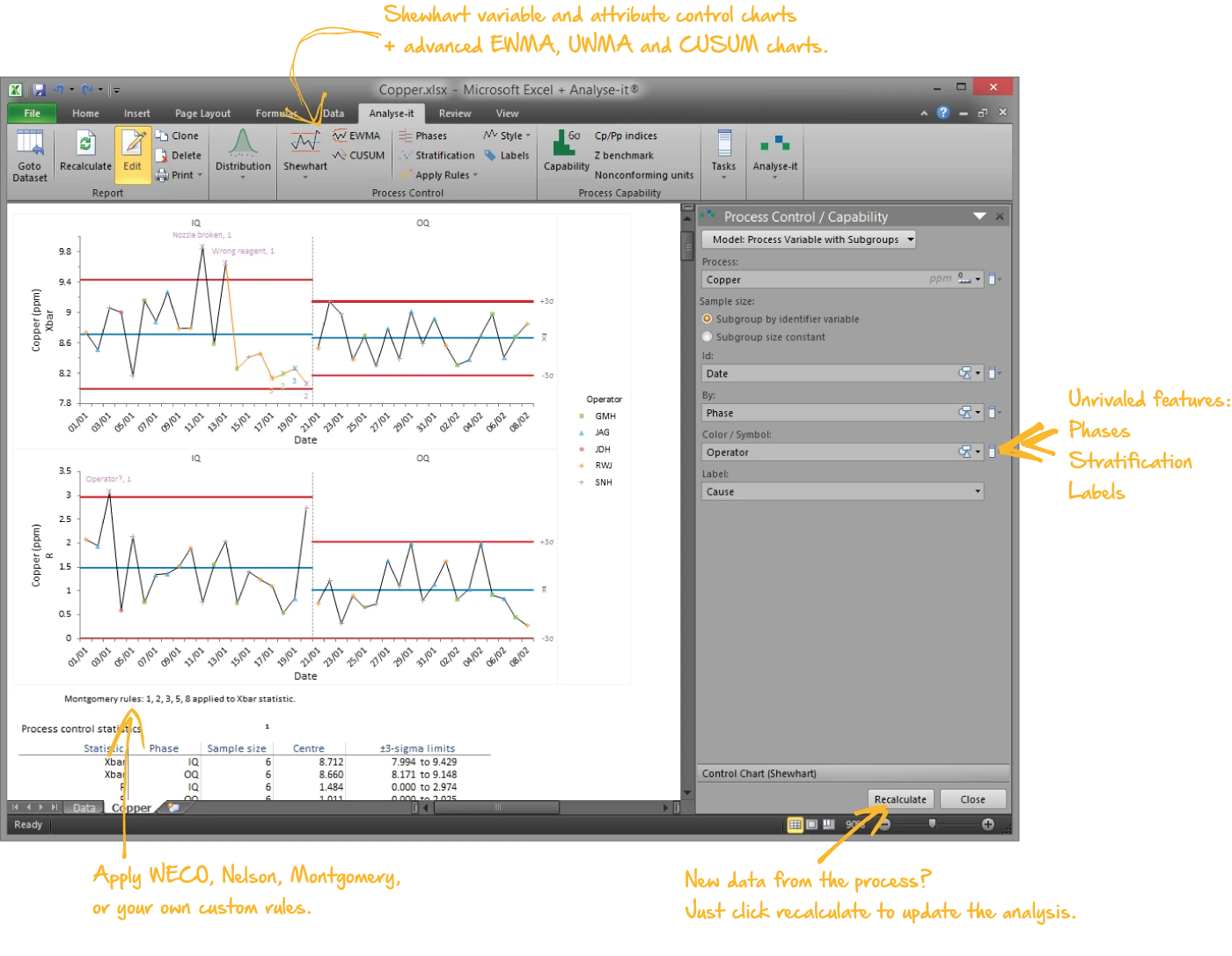
Monitor processes with control charts
Shewhart variable and attribute charts with automatic detection rules, plus time-weighted charts for detecting small, sustained shifts:
- Xbar-R, Xbar-S, I-MR for continuous data; p, np, c, u for attribute data
- CUSUM, EWMA, and UWMA time-weighted charts
- WECO, Nelson, and Montgomery detection rules — runs, trends, stratification, and oscillation flagged automatically
- Phases for before-and-after comparison with separate control limits per period
- Stratification by operator, shift, machine, material lot, or any factor
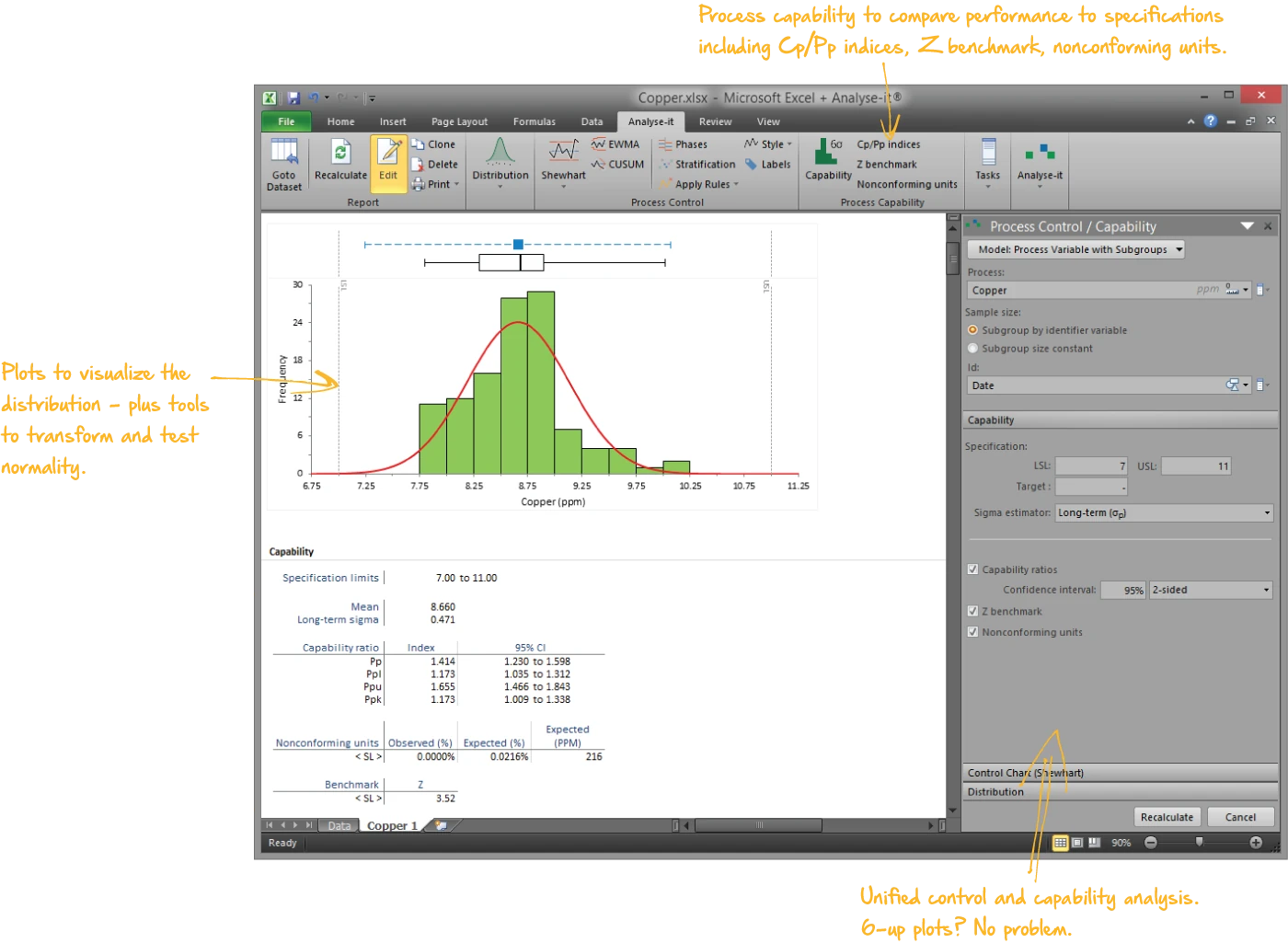
Determine whether the process meets the specification
Capability and performance indices with confidence intervals and non-normal data handling:
- Cp, Cpk, Cpm for inherent capability; Pp, Ppk for actual delivered performance
- Z-benchmark, sigma level, and nonconforming unit estimation
- Box-Cox and power transformations for non-normal distributions
- Histogram with specification limits and normal Q-Q plot with Lilliefors band
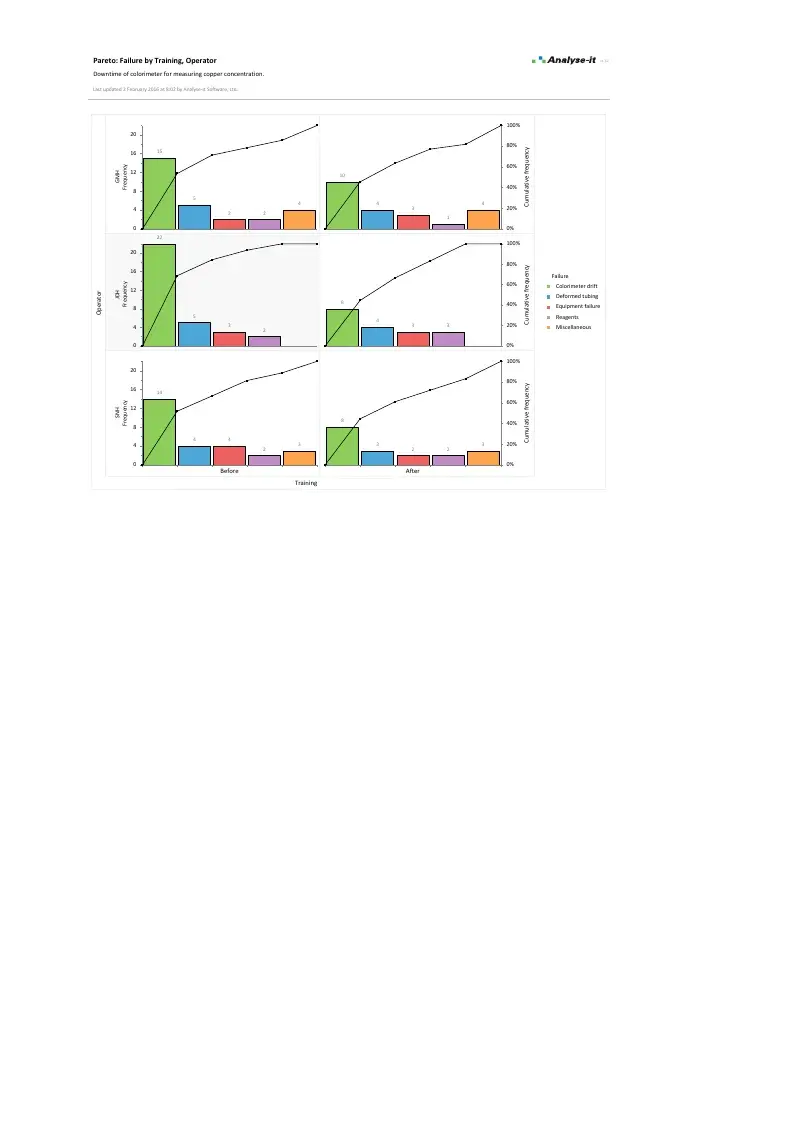
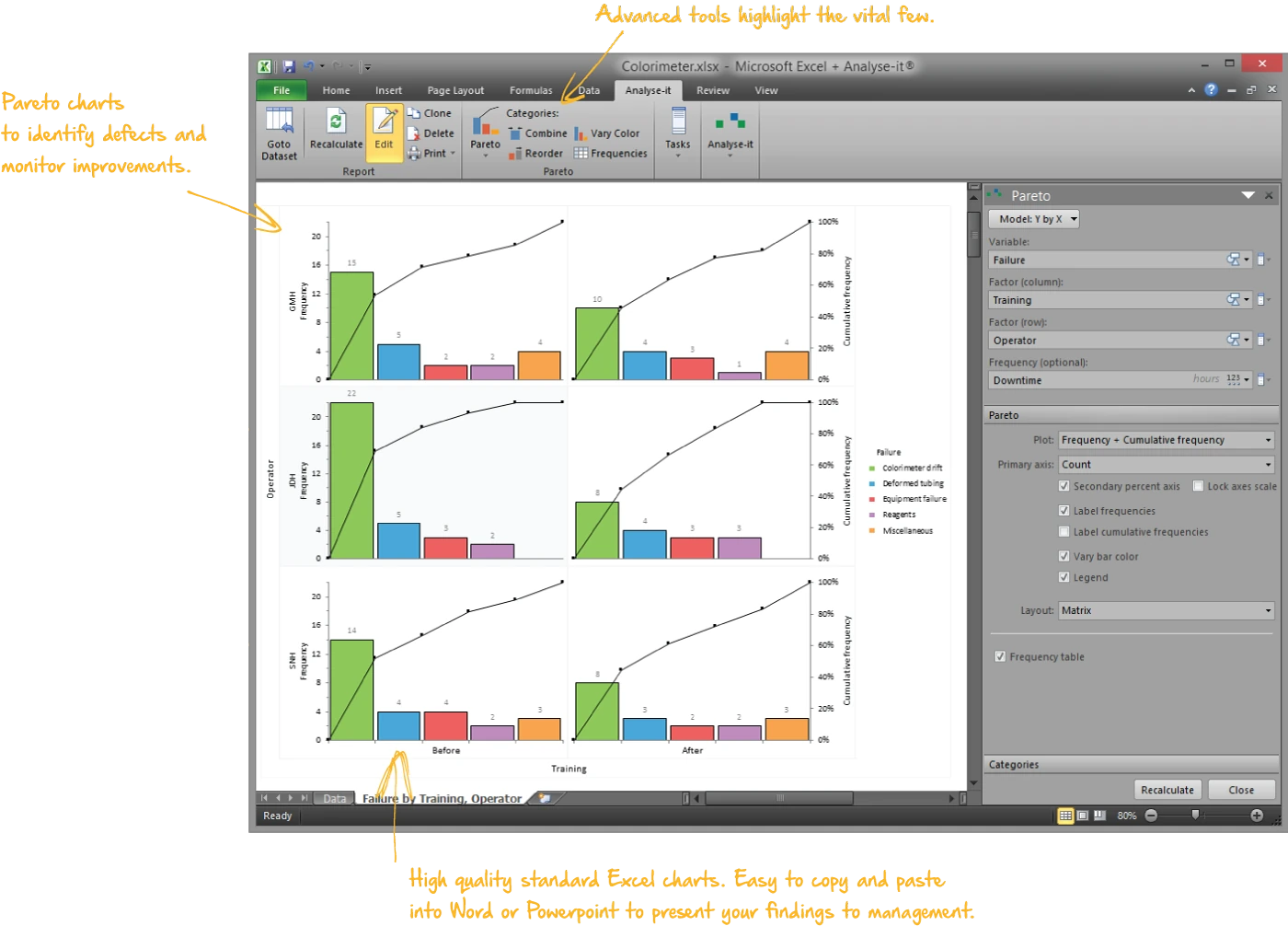
Focus improvement effort on the vital few
Pareto charts to identify the most frequently occurring defects and break them down by contributing factors:
- One-way and two-way comparative Pareto charts — defect type by operator, shift, machine, product line
- Merge low-frequency categories, reorder bars, colour by subgroup
- 80/20 cumulative percentage line
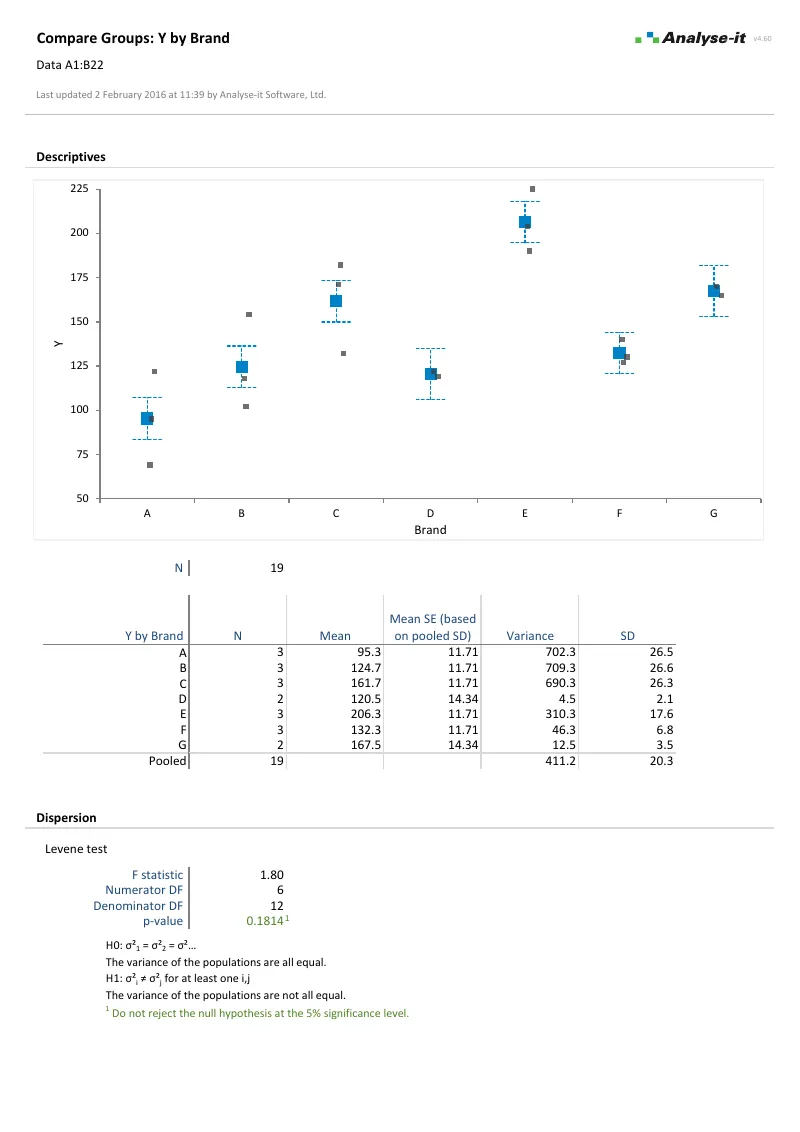
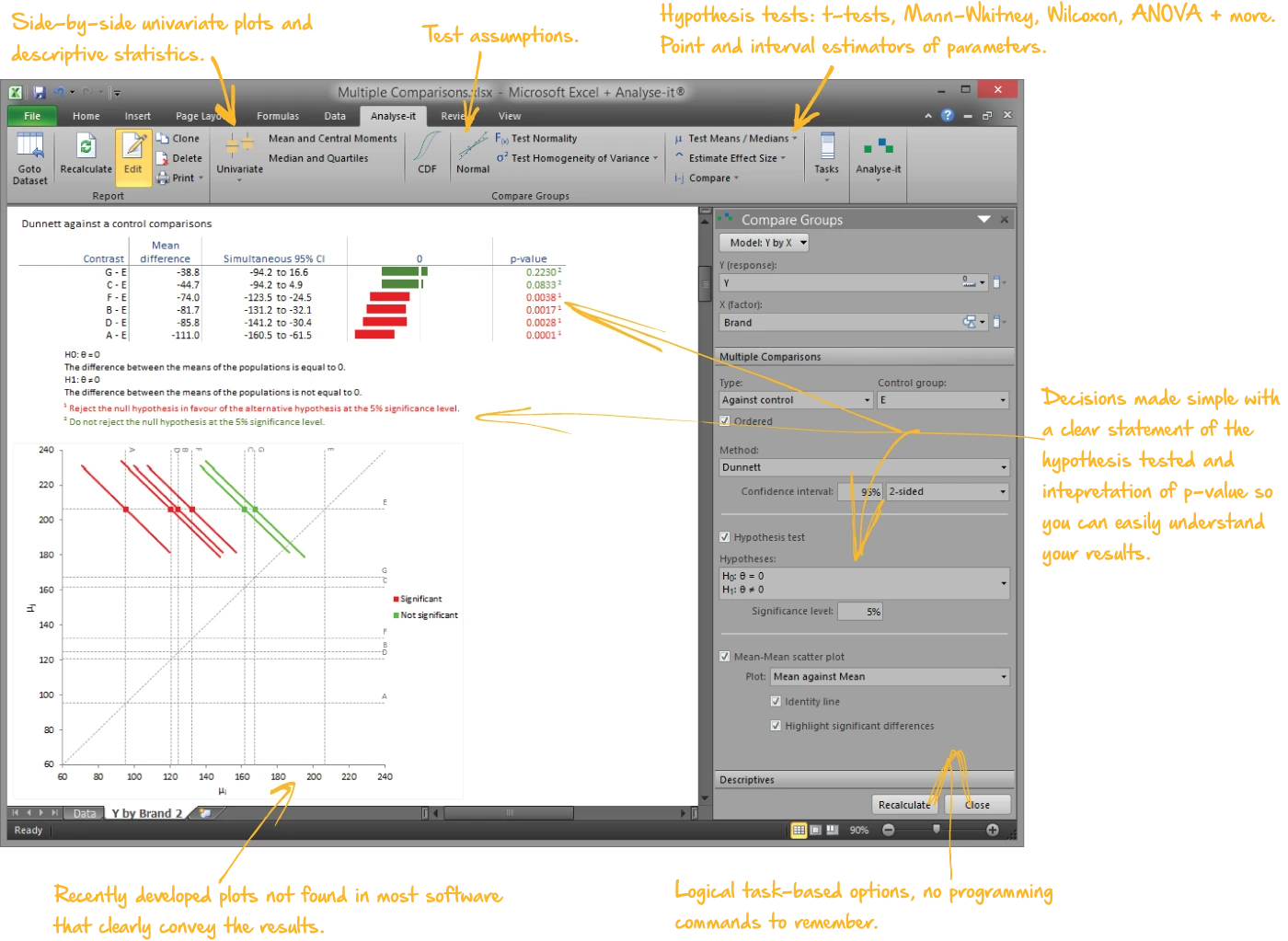
Compare groups and test for differences
Independent samples, paired samples, two groups or ten — parametric and non-parametric tests with the assumption checks built into the same workflow:
- Student’s t, Welch’s t, Wilcoxon-Mann-Whitney for two groups; one-way ANOVA, Welch’s ANOVA, Kruskal-Wallis for three or more
- Paired t-test, Wilcoxon signed ranks, Sign test, within-subjects ANOVA, Friedman
- Nine multiple comparison procedures — Tukey-Kramer, Dunnett, Hsu, Scheffé, Steel, Dwass-Steel-Critchlow-Fligner
- Cohen’s d and Hedges’ g effect sizes with non-central t confidence intervals
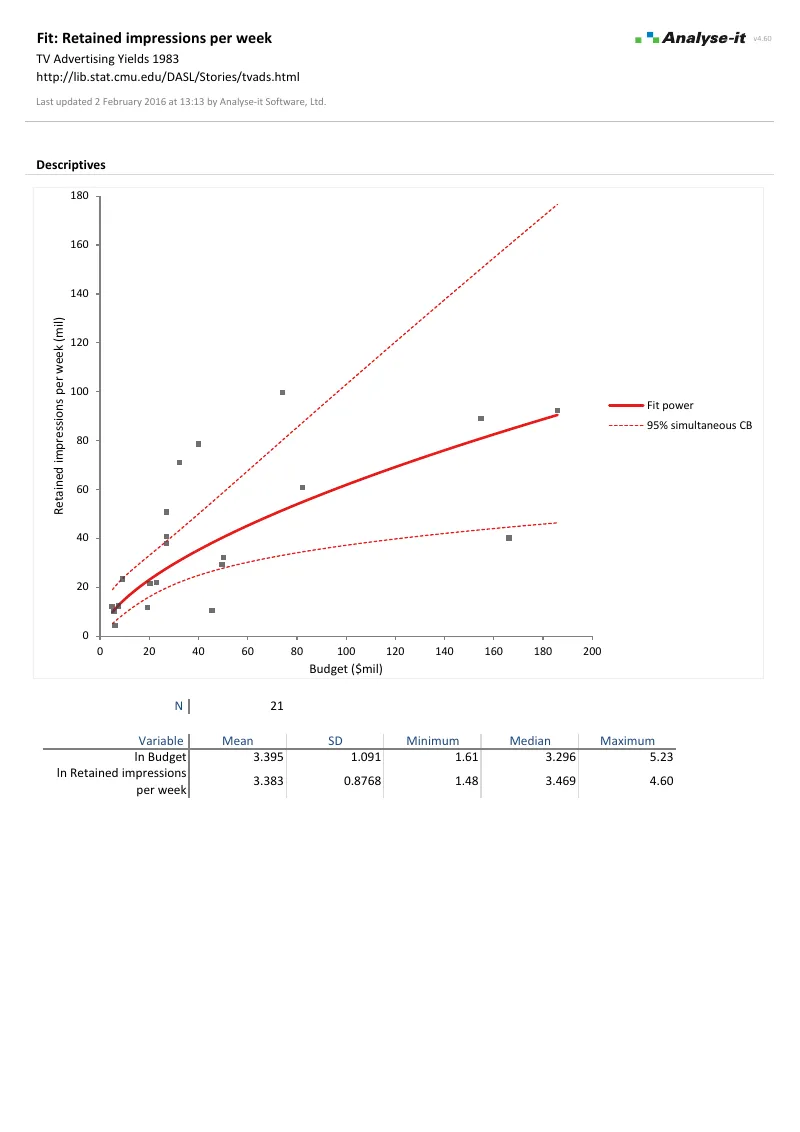
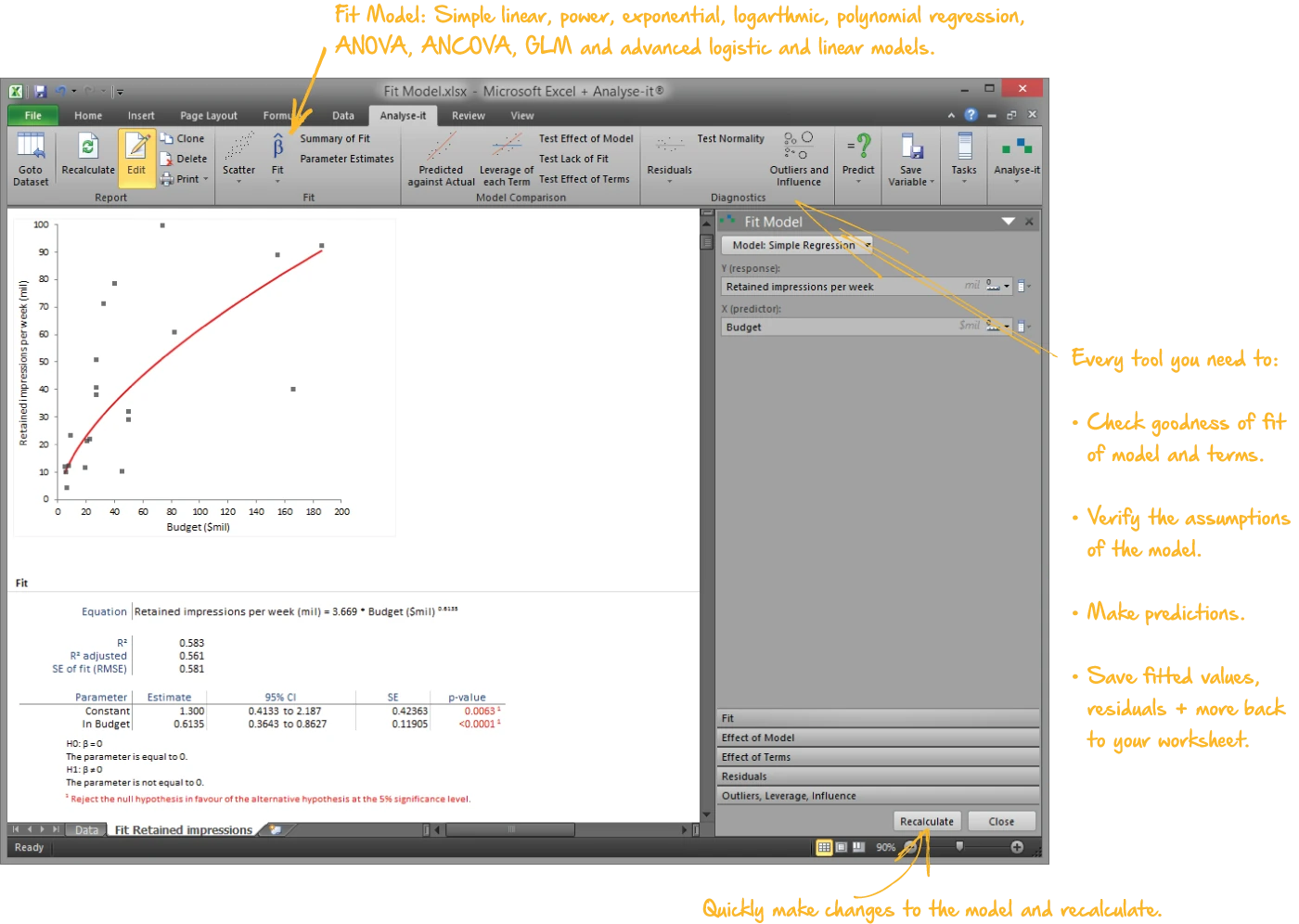
Fit and diagnose regression models
Regression as a process of building, examining, and refining — not a single pass from data to p-value:
- Simple linear, polynomial, logarithmic, exponential, power, and probit regression
- Multiple regression with continuous and categorical predictors, crossed terms, and interactions
- Binary logistic regression with odds ratios and Wald confidence intervals
- ANOVA and ANCOVA with effect means, interaction plots, and multiple comparisons
- Residual diagnostics, leverage plots, Cook’s D influence, VIF for multicollinearity
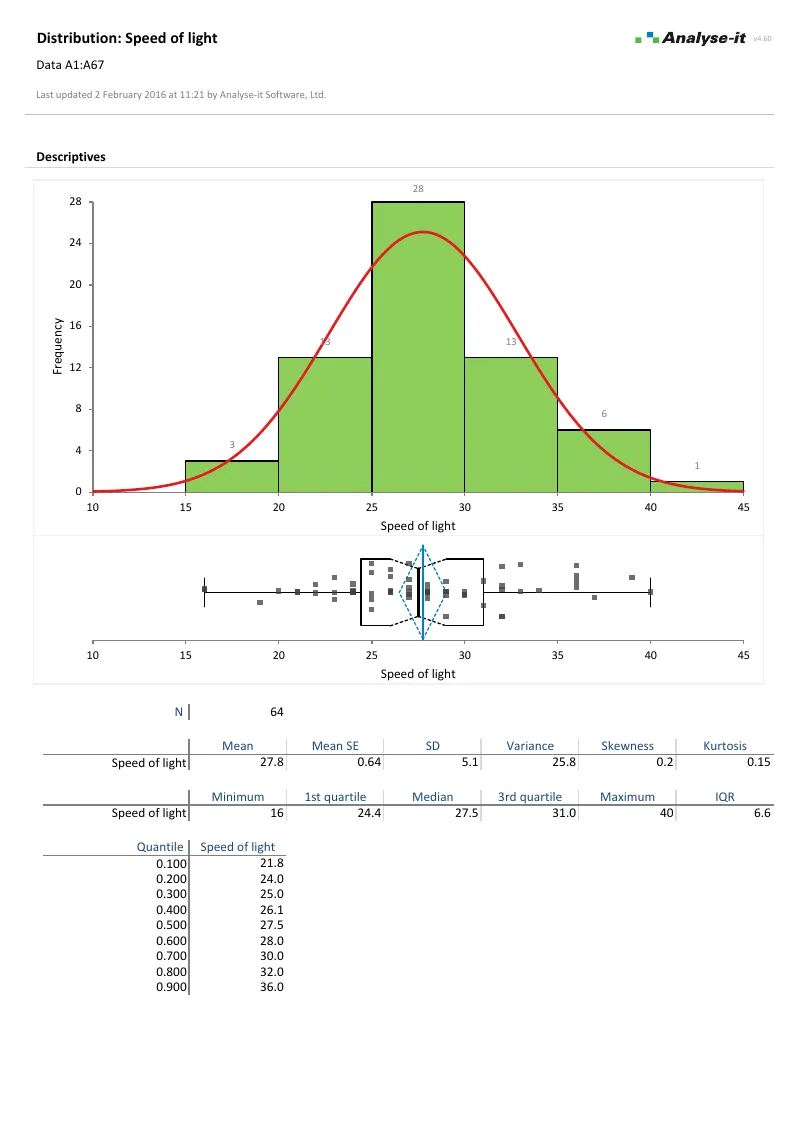
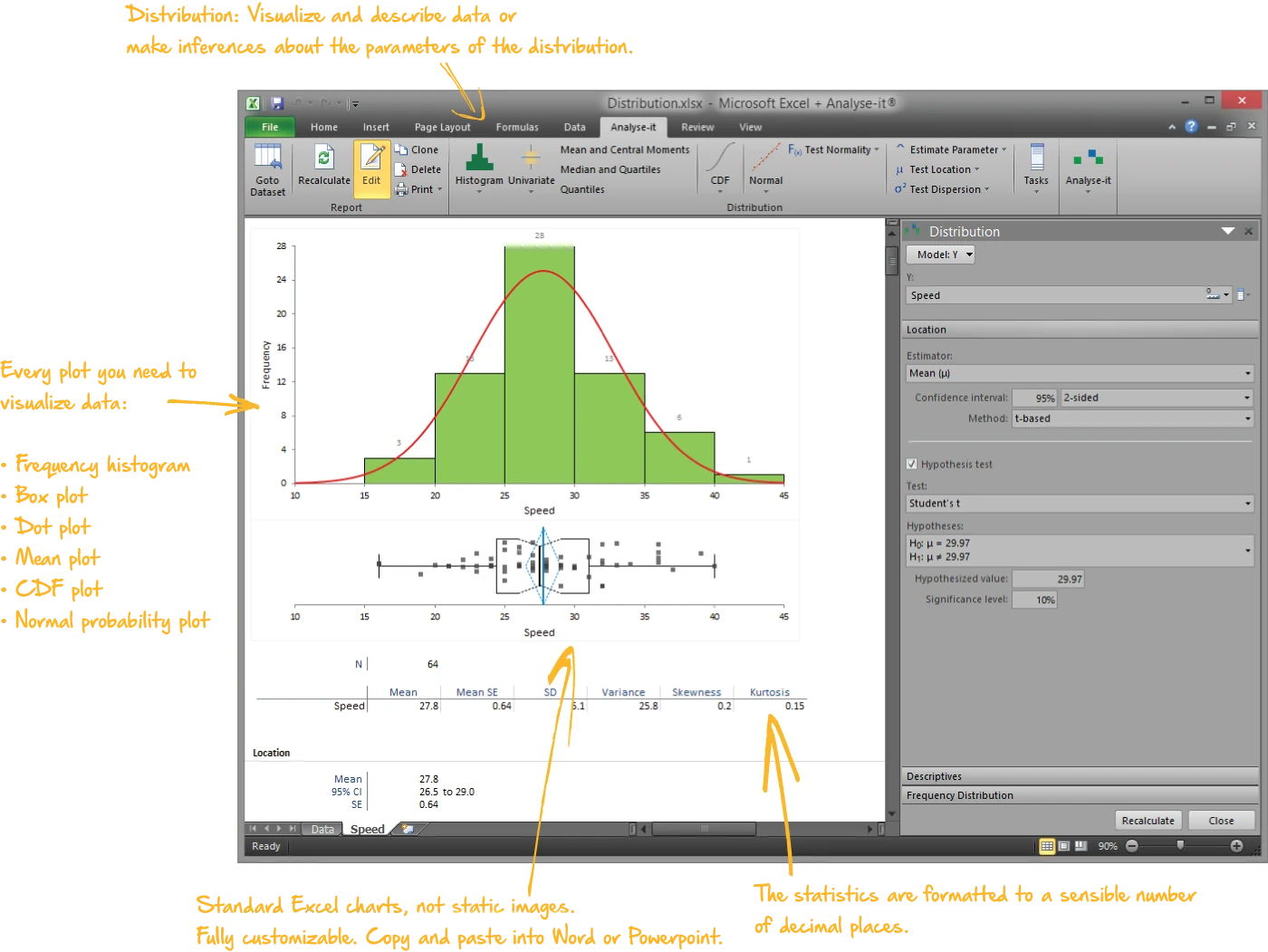
Describe and visualise data
Every analysis starts with understanding the data. What does the distribution look like? Are there outliers? Is it normal?
- Mean, median, SD, CV%, skewness, kurtosis, geometric mean, harmonic mean, quantiles, mode
- Histograms, dot plots, box plots (skeletal, Tukey outlier, quantile), CDF plots, Q-Q plots with Lilliefors bands
- Shapiro-Wilk, Anderson-Darling, and Kolmogorov-Smirnov normality tests
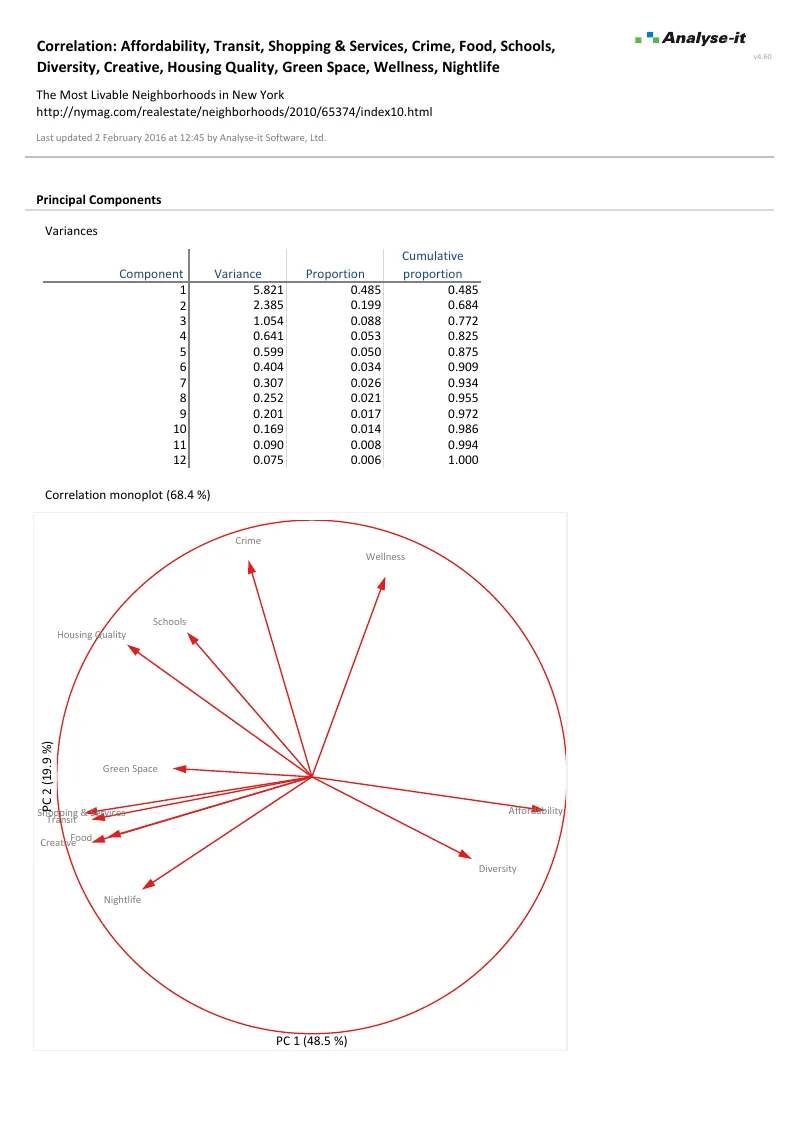
- Correlation — Pearson r, Spearman rs, Kendall τ with confidence intervals and colour-mapped matrices
- PCA and common factor analysis with Gabriel and Gower-Hand biplots, 12 rotation methods
Technical Product Manager
Alfa Wassermann Diagnostic Technologies
Everything, in one licence
The Ultimate edition includes every feature from the Method Validation, Medical, Quality Control & Improvement, and Standard editions. If your organisation needs capabilities from more than one specialist edition — or you don’t want to have to choose — Ultimate is the simpler and cheaper option. You’ve already been using every feature during the 15-day trial.
Validated, reliable, trusted for over 30 years
Support for 11 CLSI protocols
The latest Clinical and Laboratory Standards Institute (CLSI) method validation protocols are recognised by the College of American Pathologists (CAP), The Joint Commission, and the US Food and Drug Administration (FDA).
| EP05-A3 | Evaluation of Precision of Quantitative Measurement Procedures | Precision |
| EP06-A | Evaluation of the Linearity of Quantitative Measurement Procedures | Linearity |
| EP09-A3 | Measurement Procedure Comparison and Bias Estimation Using Patient Samples | Method comparison |
| EP10-A3-AMD | Preliminary Evaluation of Quantitative Clinical Laboratory Measurement Procedures | MSA |
| EP12-A2 | User Protocol for Evaluation of Qualitative Test Performance | Diagnostic performance |
| EP14-A3 | Evaluation of Commutability of Processed Samples | Method comparison |
| EP15-A3 | User Verification of Precision and Estimation of Bias | MSA |
| EP17-A2 | Evaluation of Detection Capability for Clinical Laboratory Measurement Procedures | Detection capability |
| EP21-A | Estimation of Total Analytical Error for Clinical Laboratory Methods | Method comparison |
| EP24-A2 | Assessment of the Diagnostic Accuracy of Laboratory Tests Using Receiver Operating Characteristic Curves | Diagnostic performance |
| EP28-A3C | Defining, Establishing, and Verifying Reference Intervals in the Clinical Laboratory | Reference intervals |
Clinical Chemistry Reagent Development
Beckman Coulter, Inc.
Director, Nuclear Cardiology
North Shore University Hospital
Example analyses
Download example datasets, open them in the trial, and see exactly what the output looks like.
Method Validation
Medical
Quality Control & Improvement
Standard
Technical details
The Ultimate edition includes every feature from all four editions. Full technical specifications are on each edition’s page:
- Method Validation edition — precision, linearity, detection capability, method comparison, reference intervals, diagnostic performance, 11 CLSI protocols
- Medical edition — Bland–Altman agreement, diagnostic accuracy, reference intervals, survival analysis
- Quality Control & Improvement edition — control charts, process capability, Pareto analysis
- Standard edition — descriptive statistics, hypothesis tests, ANOVA/ANCOVA, regression, logistic regression, PCA, correlation, contingency tables
System requirements
- Microsoft Excel 2013, 2016, 2019, 2021, 2024 and Microsoft 365 for Microsoft Windows (32- and 64-bit)
- Microsoft Windows 8, 10, 11, Server 2016, 2019, 2022
- 2 GB RAM minimum recommended
- 80 MB disk space