Reference interval software for method validation Establish, partition, verify, and transfer reference intervals per EP28-A3C — parametric, non-parametric, robust, bootstrap, and Harrell-Davis quantile methods with a full range of transformations.
Establish reference ranges that reflect real patient populations
Clinicians interpret every result against the reference interval. If the interval doesn’t represent the population being tested — wrong quantile method for the sample size, no partitioning for biologically distinct subgroups, non-normal data forced through a parametric calculation — the clinical interpretation is unreliable. Patients get flagged as abnormal when they’re not, or genuinely abnormal results slip through. IVD manufacturers need reference intervals for product labelling; clinical laboratories need to establish, verify, or transfer them for every analyte they report.
Analyse-it covers the full EP28-A3C workflow — five quantile methods, seven transformations, partitioning by any combination of factors, and both transfer and verification workflows. Choose the right method for the data in front of you, not the one the software happens to support.
Technical Product Manager
Alfa Wassermann Diagnostic Technologies
What's included
Five quantile methods for any distribution and sample size
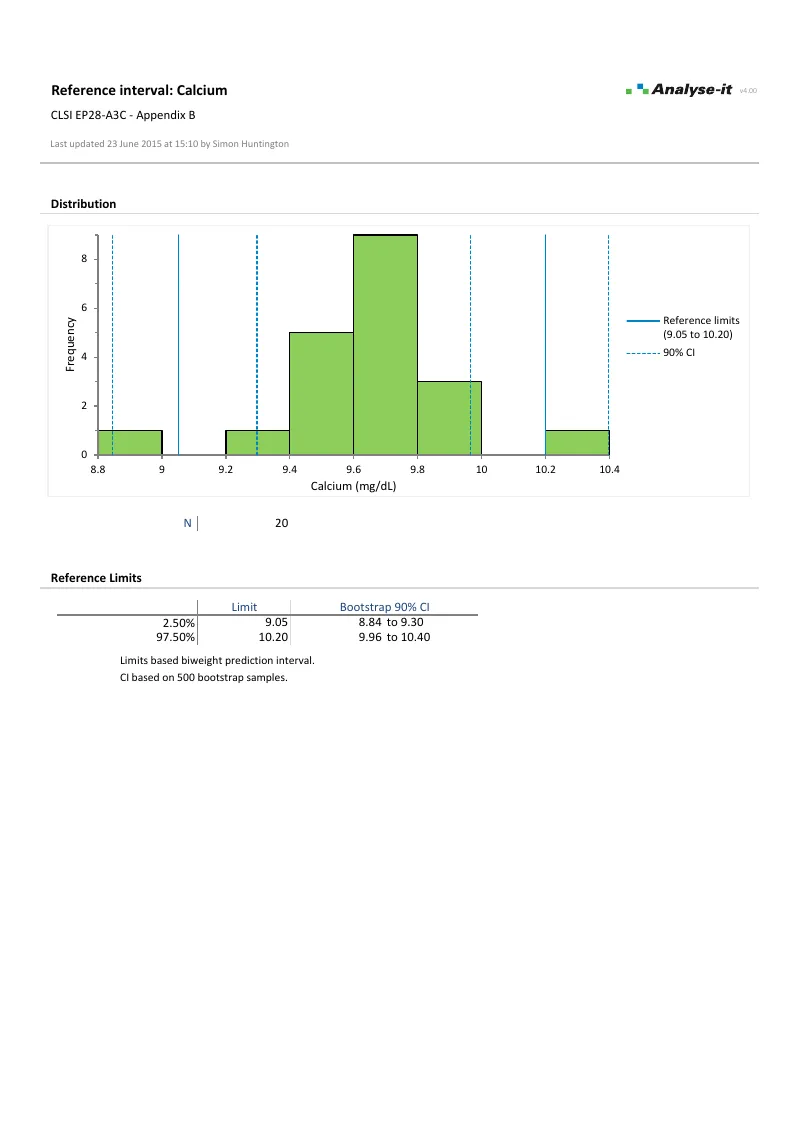
Parametric, non-parametric with three computation approaches — (N+1)p, Np+½, and (N+⅓)p+⅓ — robust bi-weight, bootstrap, and Harrell-Davis. Confidence intervals on all reference limits. Compare methods on the same dataset to assess how sensitive the limits are to the estimation approach.Partition by sex, age, ethnicity, or any combination
Partition by any combination of factors within a single analysis — such as sex and age group for an analyte like alkaline phosphatase where both matter. Each subgroup maintains its own sample, its own outlier screening, and its own reference limits.Seven transformations for non-normal distributions
Log, square root, cube root, reciprocal, Box-Cox, Manly exponential, and two-stage exponential/modulus. Shapiro-Wilk and Anderson-Darling normality tests plus normal Q-Q plots before and after transformation.Tukey outlier screening
Tukey outlier box plot to identify and flag extreme values before they distort the reference limits.Transfer and verify existing reference intervals
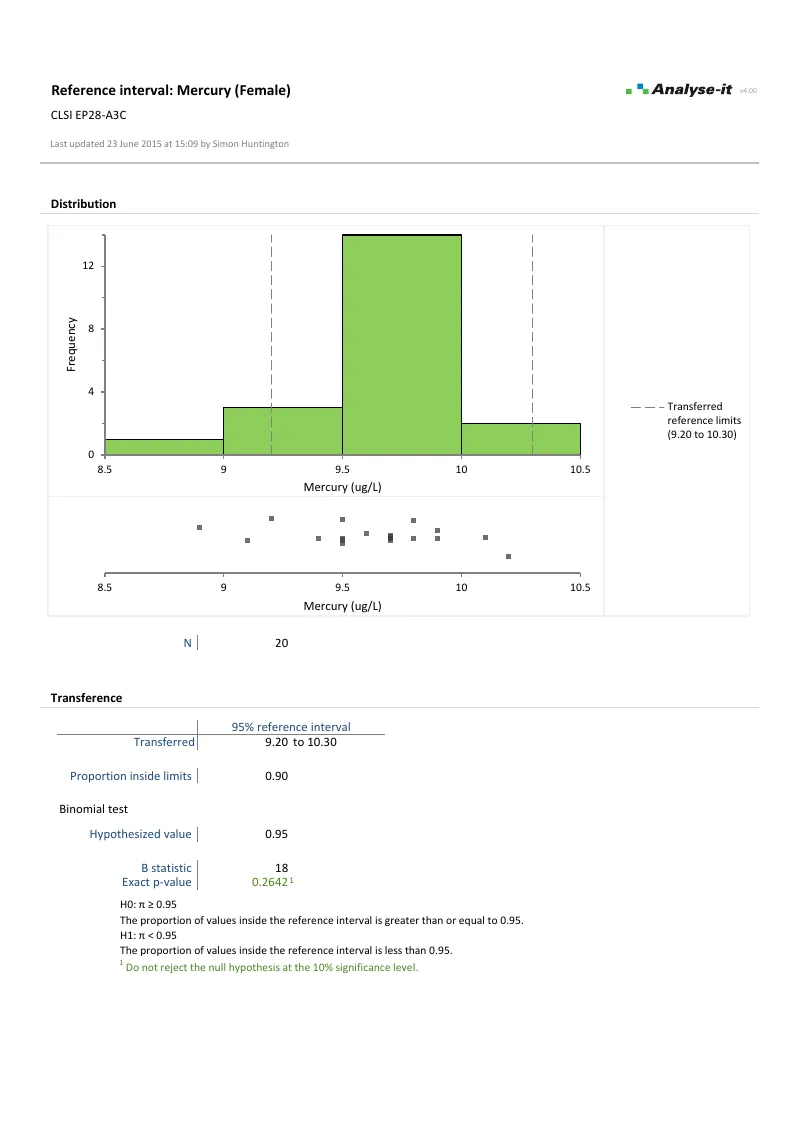
Transfer an existing interval to a new measurement procedure using the regression function from a method comparison study. Verify transferred limits using a binomial test for the proportion falling inside the reference interval — typically 20 samples per partition per EP28-A3C.
Example analyses
See reference interval results in detail — quantile methods, partitioning, transformations, and transfer/verification — using CLSI example datasets you can download and follow along with.
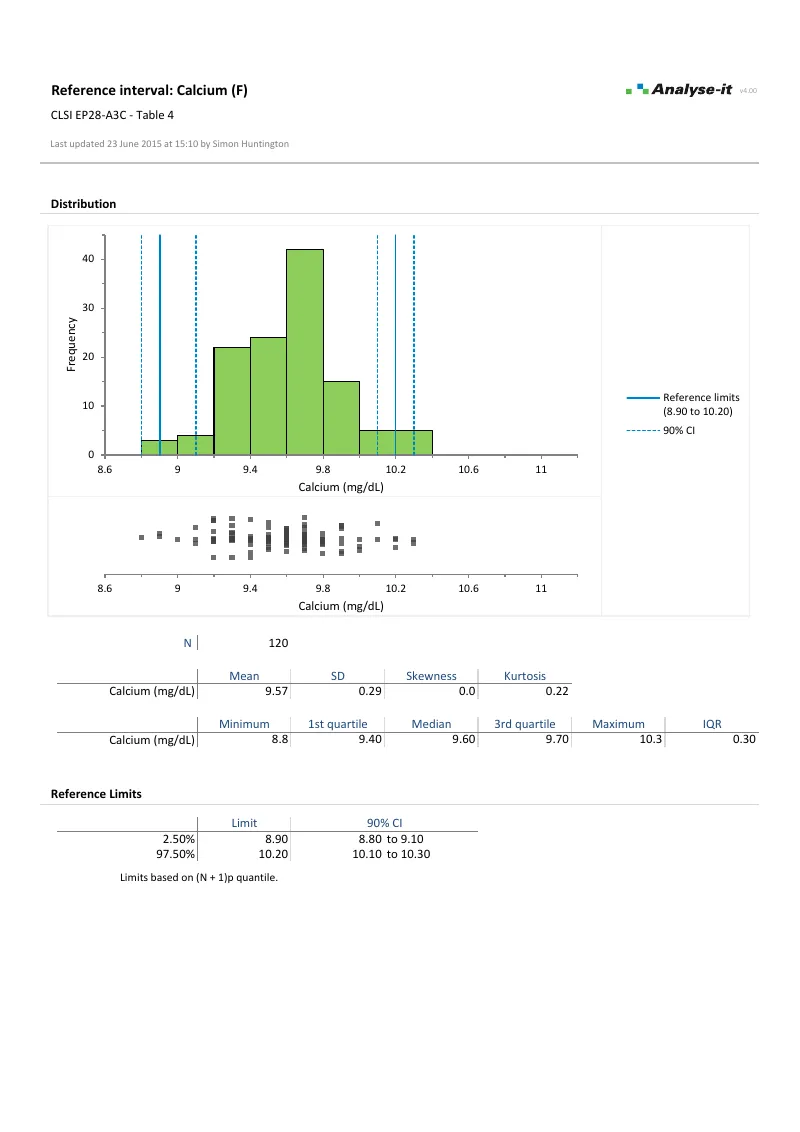 EP28-A3C — Table 4
EP28-A3C — Table 4Calcium reference intervals. Partitioned by sex. Non-parametric (N+1)p quantile method, 120 observations per group. Histogram with reference limits and 90% CIs.
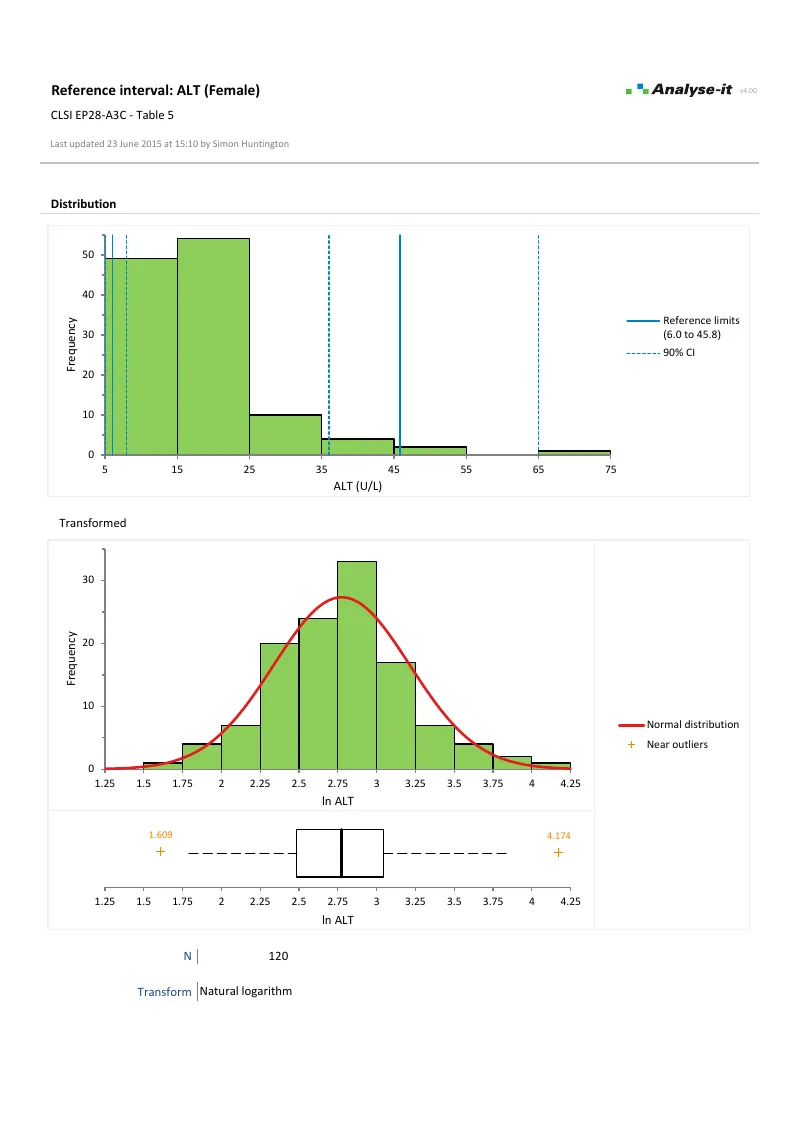 EP28-A3C — Table 5
EP28-A3C — Table 5ALT reference intervals. Partitioned by sex. Log transformation for non-normal data. Non-parametric quantile reference limits on transformed data. Histograms, Q-Q plot, and Tukey box plot.
Part of the Method Validation Edition
Reference intervals are one part of the Method Validation Edition, alongside measurement system analysis, method comparison, and diagnostic performance.
Software you can trust
Technical details
CLSI protocols
- EP28-A3C: Defining, Establishing, and Verifying Reference Intervals in the Clinical Laboratory
Establish reference limits
- Normal (parametric) quantile
- Non-parametric quantile: (N+1)p, Np+½, (N+1/3)p+1/3
- Harrell-Davis quantile
- Bootstrap quantile
- Robust bi-weight quantile
- Confidence intervals on all reference limits
Transfer / verify
- Transfer using method comparison regression function
- Binomial test for proportion inside reference interval
Partitioning & transformations
- Partition by factor(s) new in v4.00
- Reciprocal transformation
- Log transformation
- Square and cube root
- Box-Cox new in v3.52
- Manly exponential new in v4.00
- 2-stage exponential / modulus new in v4.00
Normality testing
- Shapiro-Wilk test
- Anderson-Darling test
- Normal Q-Q plot
Plots
- Frequency distribution histogram with normal overlay and reference limits
- Tukey outlier box plot
- Normal Q-Q plot with Lilliefors confidence band