Measurement system analysis software for method validation Precision, trueness, linearity, interferences, and detection capability — five CLSI protocols in one analysis for characterising measurement system performance.
Characterise the full performance of a measurement procedure in one analysis
Before a measurement procedure goes into clinical use, you need to know how precise it is, how accurate, whether it’s linear across the reportable range, how susceptible it is to common interferents, and the lowest concentration it can reliably detect. Each characteristic has its own CLSI protocol, its own experimental design, and its own acceptance criteria. Doing this work in separate tools means re-entering data, reconciling formats, and risking errors at every handoff — especially problematic when the results feed into a 510(k) submission or accreditation file.
Analyse-it unifies all five protocols in one analysis. Design the experiment with flexible nested factors, run precision, trueness, linearity, interferences, and detection capability, and let precision profiles feed directly into LoB, LoD, and LoQ estimation — no re-entering data, no separate tools.
Clinical Chemistry Reagent Development
Beckman Coulter, Inc.
What's included
Establish or verify precision →
Variance component analysis with flexible nested designs per EP05-A3. Separate repeatability from between-run, between-day, between-operator, and between-site contributions. Precision profiles show how imprecision changes across the measuring range — with six variance function models that feed directly into detection capability estimation.Confirm linearity across the measuring range →
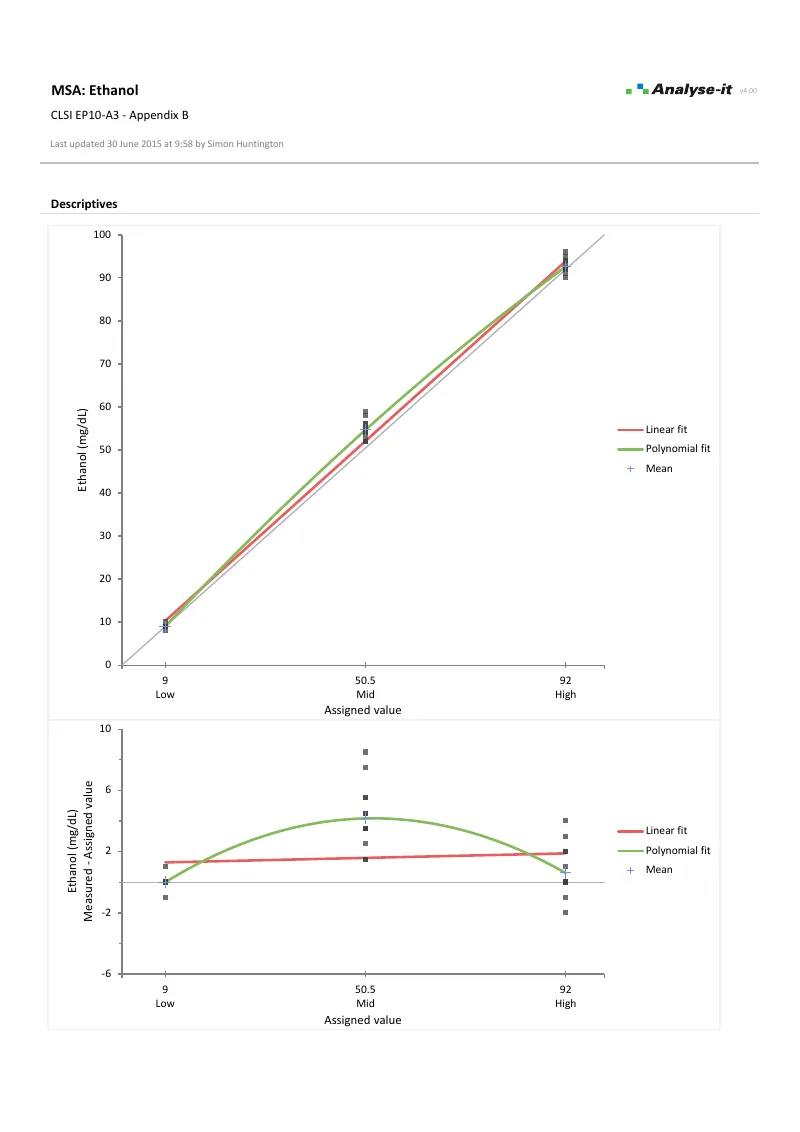
Polynomial regression with nonlinearity testing per EP06-A. Determine where the assay is linear, adjust the measuring interval to find the reportable range, and test whether nonlinearity is within allowable limits — such as ±5% — using Hsieh-Liu confidence intervals. Weighted models for non-constant precision.Establish detection limits →
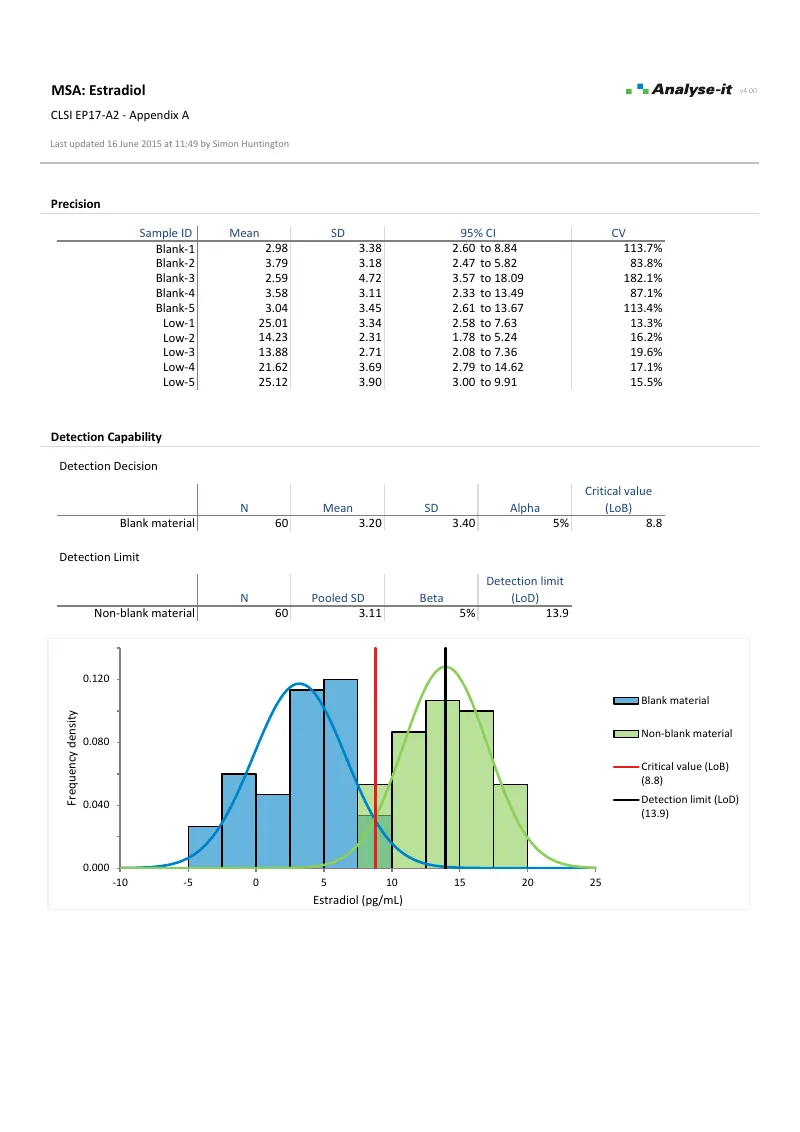
LoB, LoD, and LoQ estimation per EP17-A2 using parametric, non-parametric, probit regression, and precision profile variance function approaches. Precision profiles from EP05-A3 feed straight in.Verify precision and trueness against manufacturer claims
Pass/fail verification per EP15-A3. Test precision against the claimed CV using a chi-squared test, and estimate bias against assigned values with equality and equivalence tests — such as “bias within ±2 mg/dL of the assigned value.” Difference plot with allowable bias bands.Evaluate interferences per EP10-A3-AMD
Estimate the magnitude of interference from haemolysis, lipaemia, bilirubin, medications, or other substances and test whether it exceeds an allowable limit at clinically relevant concentrations.Flexible experiment design with outlier detection
Up to three random nested factors and one fixed factor in any arrangement, balanced or unbalanced — such as 3 sites × 5 days × 2 runs × 3 replicates, or a simpler single-lab 20 × 2 × 2 verification design. Generalised ESD outlier identification. Variability of measurements plot for visual inspection.

Example analyses
See measurement system analysis results in detail — precision, trueness, linearity, interferences, and detection capability — using CLSI example datasets you can download and follow along with.
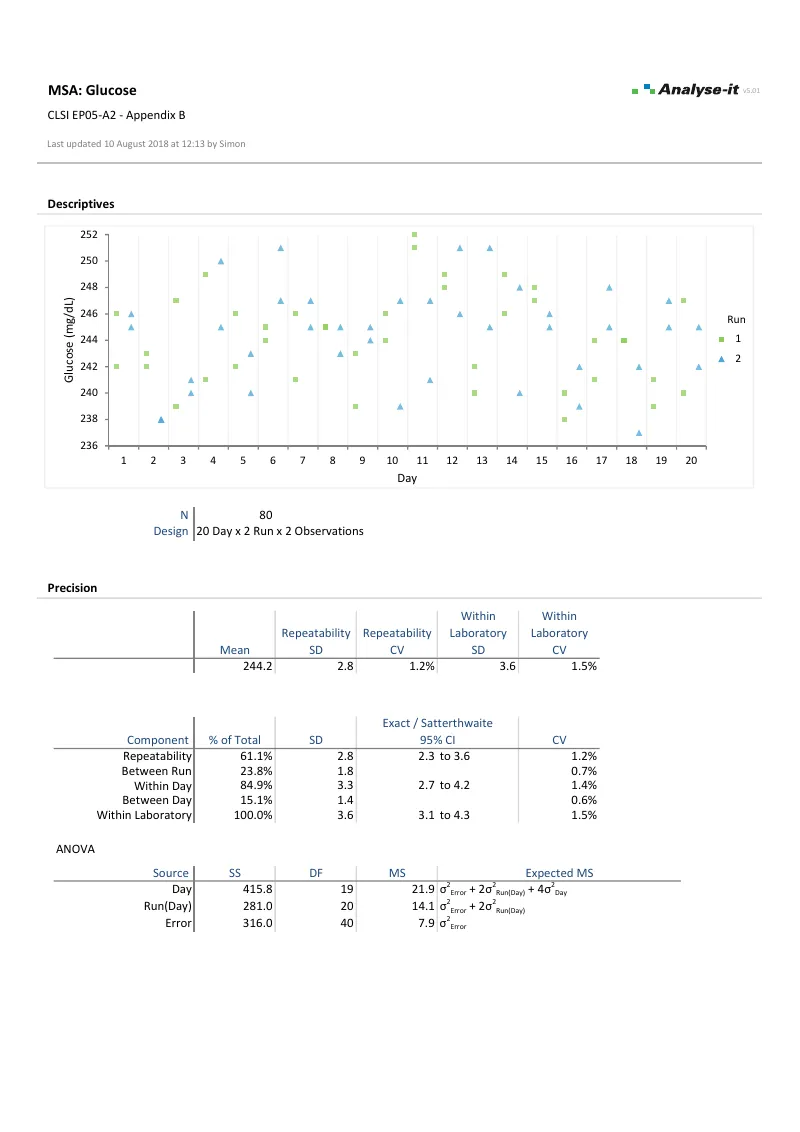 EP05-A3 — Appendix B
EP05-A3 — Appendix BGlucose precision study. 20 days × 2 runs × 2 observations. Repeatability and between-run/between-day variance components.
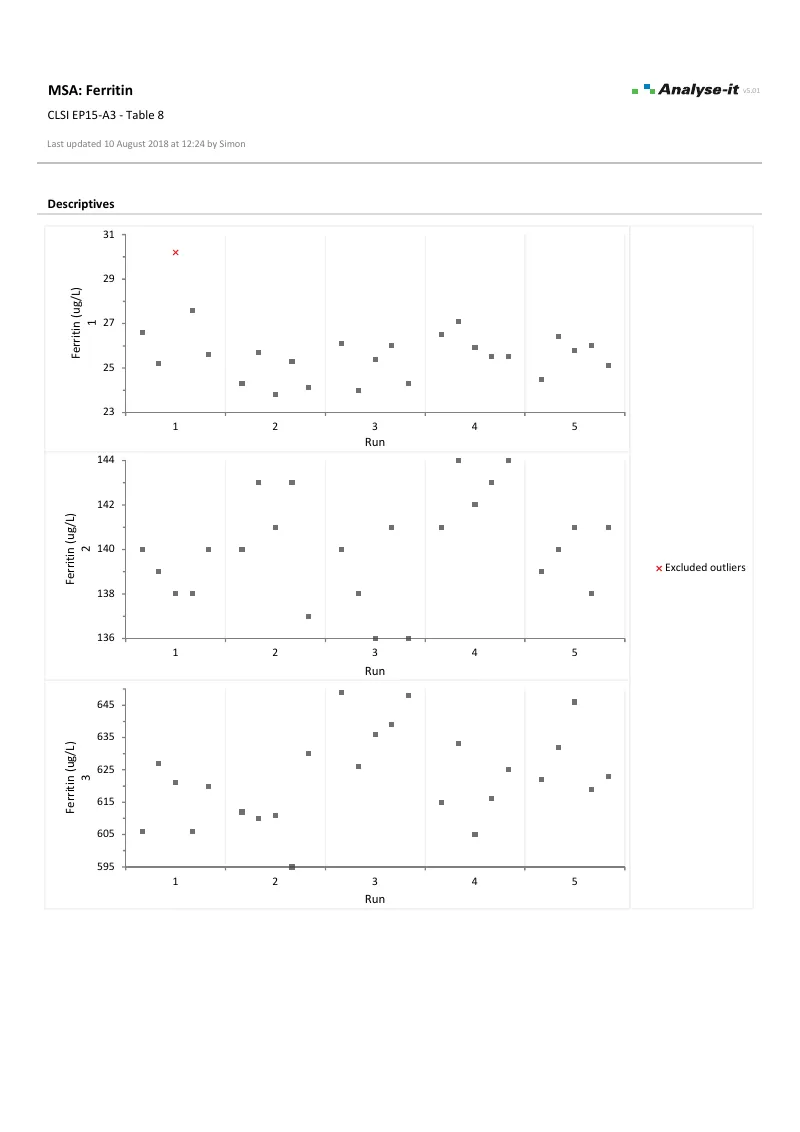 EP15-A3 — Table 8
EP15-A3 — Table 8Ferritin precision and trueness verification. 3 samples × 5 runs × 5 observations. Within-run and total precision with χ² tests against manufacturer’s claimed imprecision.
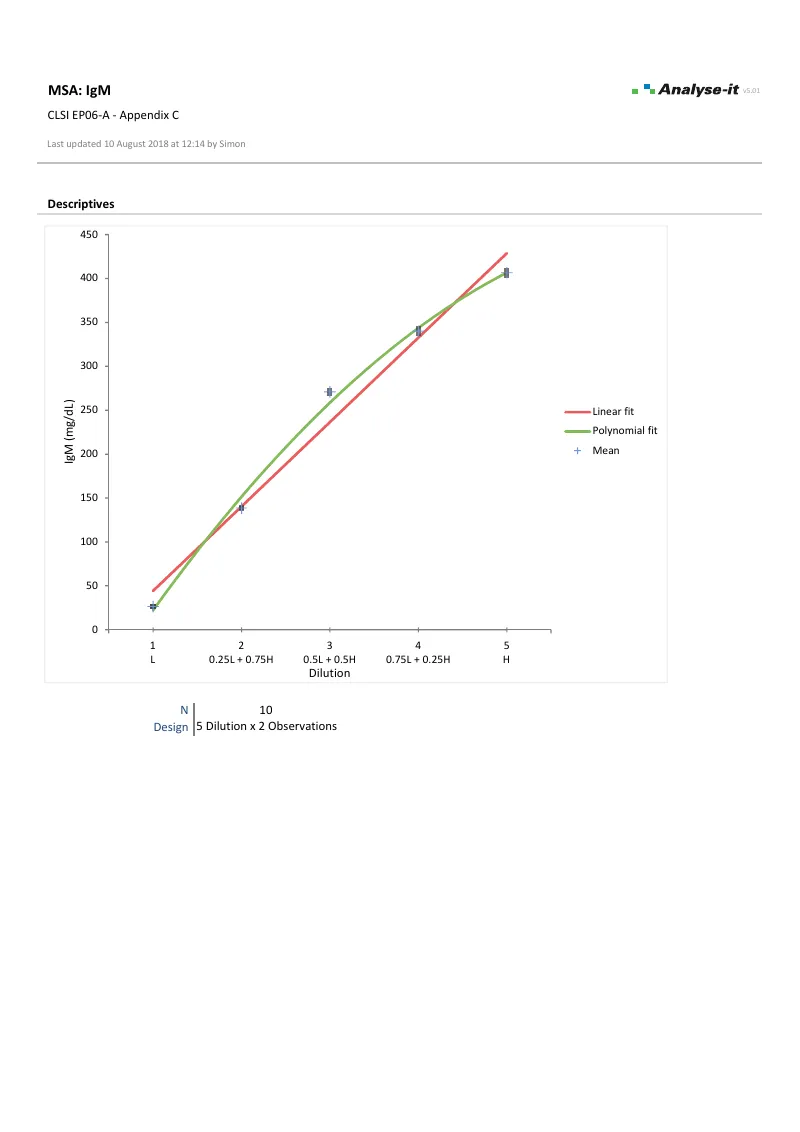 EP06-A — Appendix C
EP06-A — Appendix CIgM linearity evaluation. 5 dilutions × 2 observations. Linear and 2nd order polynomial fits with difference plot.
Part of the Method Validation Edition
Measurement system analysis is one part of the Method Validation Edition, alongside method comparison, reference intervals, and diagnostic performance.
Software you can trust
Technical details
CLSI protocols
- EP05-A3: Evaluation of Precision of Quantitative Measurement Procedures
- EP06-A: Evaluation of the Linearity of Quantitative Measurement Procedures
- EP10-A3-AMD: Preliminary Evaluation of Quantitative Clinical Laboratory Measurement Procedures
- EP15-A3: User Verification of Precision and Estimation of Bias
- EP17-A2: Evaluation of Detection Capability for Clinical Laboratory Measurement Procedures
Experiment design
- Up to 3 random nested factors new in v4.60
- 1 fixed factor (e.g. level)
- Balanced and unbalanced designs
Precision
- Precision as variance, SD, or CV%
- Exact, Satterthwaite, and MLS confidence intervals
- Abbreviated reproducibility/repeatability and detailed intermediate precision components
- ANOVA table
- Terminology: total/within, reproducibility/repeatability, or laboratory/repeatability
- χ² test against precision claim
Precision profiles
- SD or CV across the measuring range
- Constant variance function
- Constant CV function
- Mixed constant/proportional variance function
- Sadler 3-parameter power function new in v4.00
- 3-parameter alternative power function new in v4.00
- 4-parameter function with turning point new in v4.91
Bias / trueness new in v4.00
- Bias with confidence interval
- Difference plot of bias against assigned values with allowable bias bands
- Test equality (no bias) or equivalence (bias within allowable bias)
Linearity
- Linear, polynomial (2nd to 5th order), forward stepwise, and best (2nd or 3rd) polynomial regression
- Weighted models for non-constant precision new in v5.00
- Adjustable measuring interval to find linear range new in v5.00
- Difference between linear and nonlinear fit with Hsieh-Liu confidence intervals
- Equality and equivalence tests against allowable nonlinearity
- Difference plot with allowable nonlinearity band
- Emancipator-Kroll linearity
Detection capability new in v4.00
- Limit of blank (LoB): parametric (SD) or non-parametric (quantile), or from precision profile variance function
- Limit of detection (LoD): pooled SD of non-blank materials, or from precision profile variance function
- Limit of detection (LoD) using probit regression new in v5.50
- Limit of quantitation (LoQ) from precision profile variance function
Diagnostics
- Generalised ESD outlier identification new in v4.95
Plots
- Scatter plot and difference plots
- Variability of measurements plot
- Precision profile with variance function fit
- Linearity plot with linear and polynomial fits
- Difference plot with allowable nonlinearity band
- Difference plot of bias with allowable bias bands
- Frequency density histogram with LoB and LoD
- Probit regression curve new in v5.50