Precision analysis software for method validation Variance component analysis with flexible nested designs per EP05-A3 — repeatability, intermediate precision, and precision profiles across the measuring range.
Separate the sources of variability in your measurement procedure
Knowing the total imprecision isn’t enough. A method with high overall CV might have excellent within-run repeatability but large between-day drift — a different problem from one that’s noisy within every run. Without separating the variance components, you can’t identify the dominant source, you can’t take targeted corrective action, and you can’t produce the precision data that regulatory submissions and accreditation inspections require.
Analyse-it supports the full EP05-A3 protocol with flexible nested designs that match the way your laboratory actually operates. Variance components are separated at every level, precision profiles show how imprecision behaves across the measuring range, and the results feed directly into detection capability estimation and total analytical error calculations.
Clinical Chemistry Reagent Development
Beckman Coulter, Inc.
What's included
Flexible experiment design
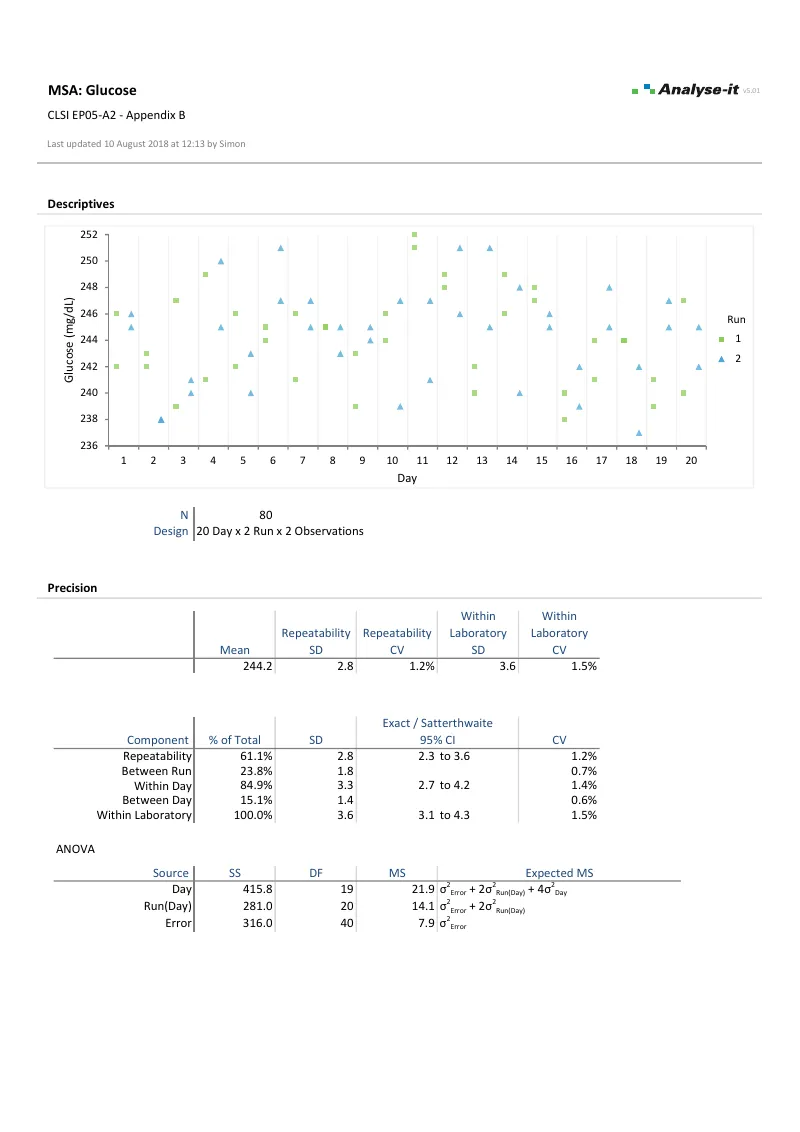
Up to three random nested factors (days, runs, operators, sites, lots) plus one fixed factor (concentration level) in any arrangement, balanced or unbalanced — such as 3 sites × 5 days × 2 runs × 3 replicates for an IVD development study, or a simpler 20 × 2 × 2 single-lab verification design.Variance components separated at every level
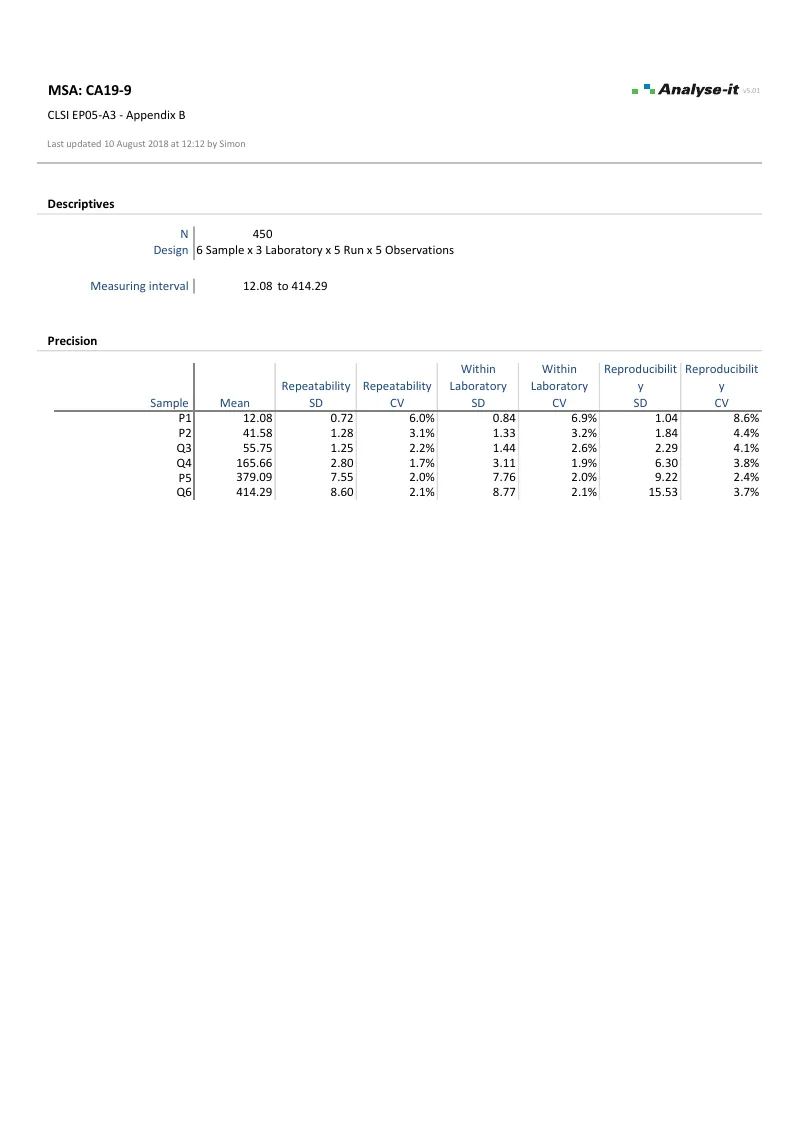
Repeatability, between-run, between-day, between-operator, between-site, within-laboratory, and reproducibility — each reported as an individual variance component with ANOVA table. Precision as variance, SD, or CV% with exact, Satterthwaite, and modified large-sample (MLS) confidence intervals.Precision profiles across the measuring range
Precision profile plots SD or CV as a function of concentration with a fitted variance function. Six models: constant variance, constant CV, mixed constant/proportional, Sadler 3-parameter power, 3-parameter alternative power, and 4-parameter function with turning point. The fitted variance function feeds directly into detection capability estimation (LoB, LoD, LoQ) via EP17-A2.Chi-squared test against precision claim
Test observed precision against a precision goal — such as the manufacturer’s claimed CV of 3.5% — for a formal pass/fail assessment per EP05-A3.Generalised ESD outlier identification
Grubbs’s test extended for multiple outliers. Variability of measurements plot for visual inspection of drift, shifts, or individual aberrations across the dataset.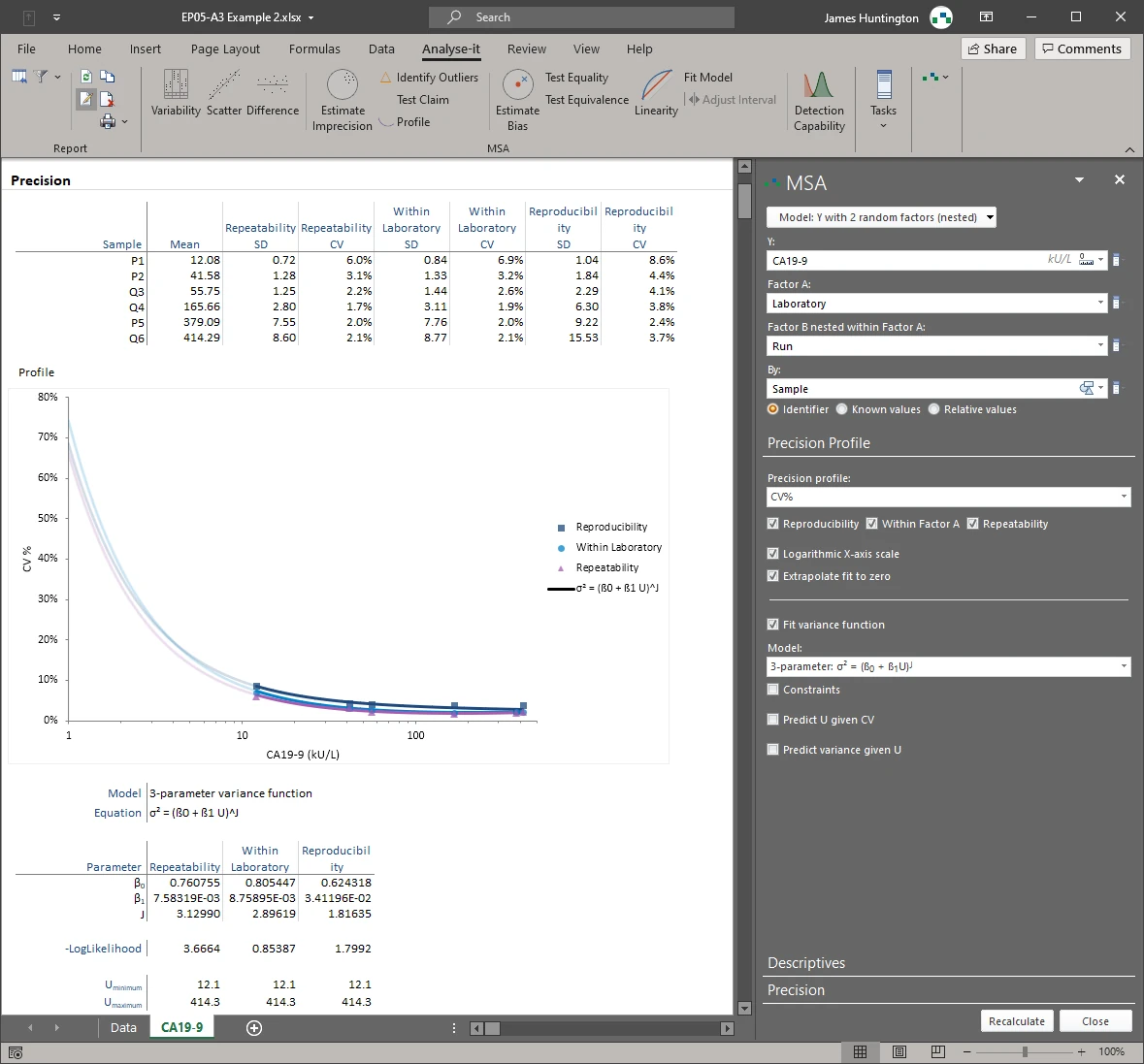
Example analyses
See precision study results in detail — variance components, ANOVA tables, precision profiles, and verification tests — using CLSI example datasets you can download and follow along with.
Part of measurement system analysis
Precision is one part of measurement system analysis, alongside linearity (EP06-A), bias/trueness verification (EP15-A3), interference evaluation (EP10-A3-AMD), and detection capability (EP17-A2). Precision profiles from EP05-A3 feed directly into detection capability estimation.
See the full MSA workflow on the measurement system analysis page →
Software you can trust
Technical details
CLSI protocols
- EP05-A3: Evaluation of Precision of Quantitative Measurement Procedures
Experiment design
- Up to 3 random nested factors new in v4.60
- 1 fixed factor (e.g. level)
- Balanced and unbalanced designs
Variance components
- Precision as variance, SD, or CV%
- Exact, Satterthwaite, and MLS confidence intervals
- Abbreviated reproducibility/repeatability and detailed intermediate precision components
- ANOVA table
- Terminology: total/within, reproducibility/repeatability, or laboratory/repeatability
Precision profiles
- SD or CV across the measuring range
- Constant variance function
- Constant CV function
- Mixed constant/proportional variance function
- Sadler 3-parameter power function new in v4.00
- 3-parameter alternative power function new in v4.00
- 4-parameter function with turning point new in v4.91
Tests & diagnostics
- χ² test against precision claim
- Generalised ESD outlier identification new in v4.95
Plots
- Variability of measurements plot
- Precision profile with variance function fit
- Scatter plot
- Difference plot