Detection capability software for method validation LoB, LoD, and LoQ estimation per EP17-A2 — parametric and non-parametric approaches, probit regression, and precision profile variance function methods.
Know what your assay can and can’t detect
Low-concentration results drive clinical decisions — ruling out disease, screening, monitoring drug levels. If the measurement procedure can’t reliably distinguish a true signal from blank noise, you risk reporting false positives or missing genuine low-level results. Getting detection limits wrong means either under-reporting (clinically dangerous) or over-reporting (eroding trust in the assay). EP17-A2 defines the framework: LoB characterises the noise floor, LoD confirms the lowest concentration you can reliably detect, and LoQ determines where measurement uncertainty is small enough for the result to be clinically actionable.
Analyse-it covers the full EP17-A2 workflow with multiple estimation approaches for each limit. Precision profiles from EP05-A3 studies feed directly into the variance function methods — no re-entering data, no separate tools for LoB, LoD, and LoQ.
Principal Scientist, Product Development
Biosite / Inverness Medical Innovations
What's included
Limit of blank from blank material or precision profile
Parametric LoB from the SD of blank measurements, non-parametric from the quantile, or from the precision profile variance function when an EP05-A3 study is already available. Frequency density histogram with LoB and LoD lines overlaid.Limit of detection from low-level samples or probit regression
LoD from pooled SD of non-blank materials per EP17-A2, from the precision profile variance function, or via probit regression for molecular and immunoassay methods where detection is probabilistic. Probit determines the LoD at the concentration where detection probability reaches the required threshold — such as 95%.Limit of quantitation from precision profile
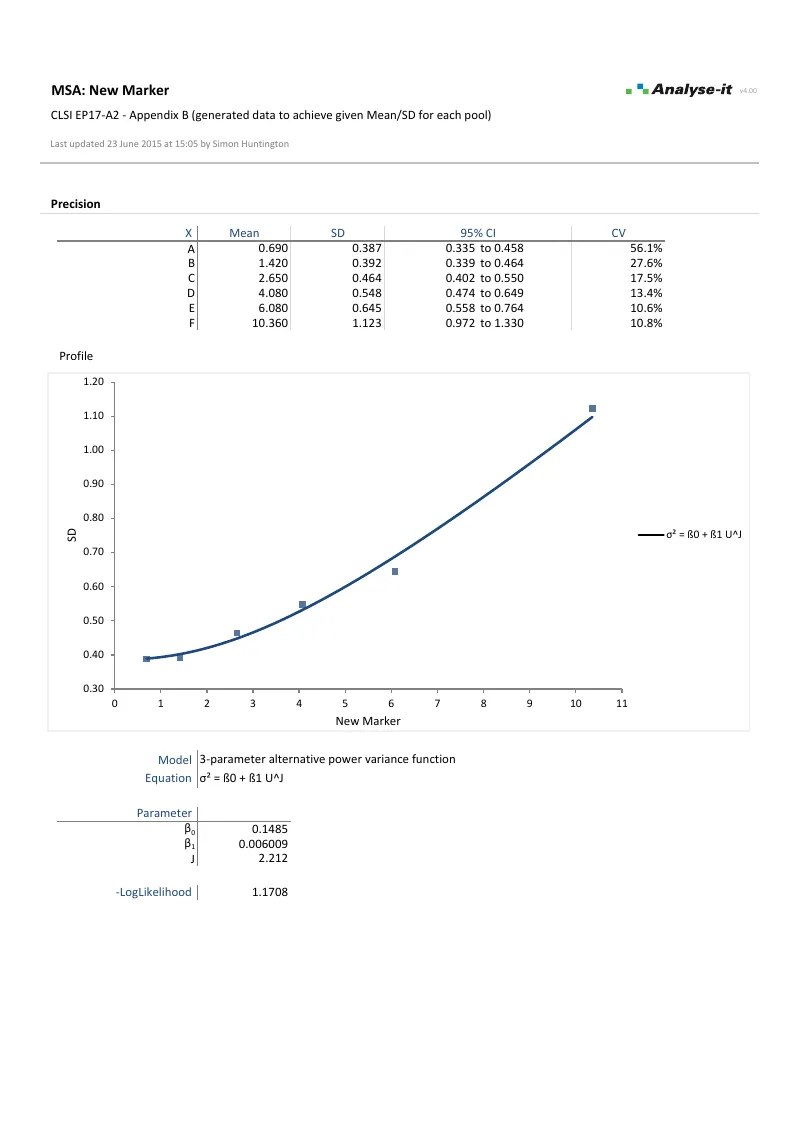
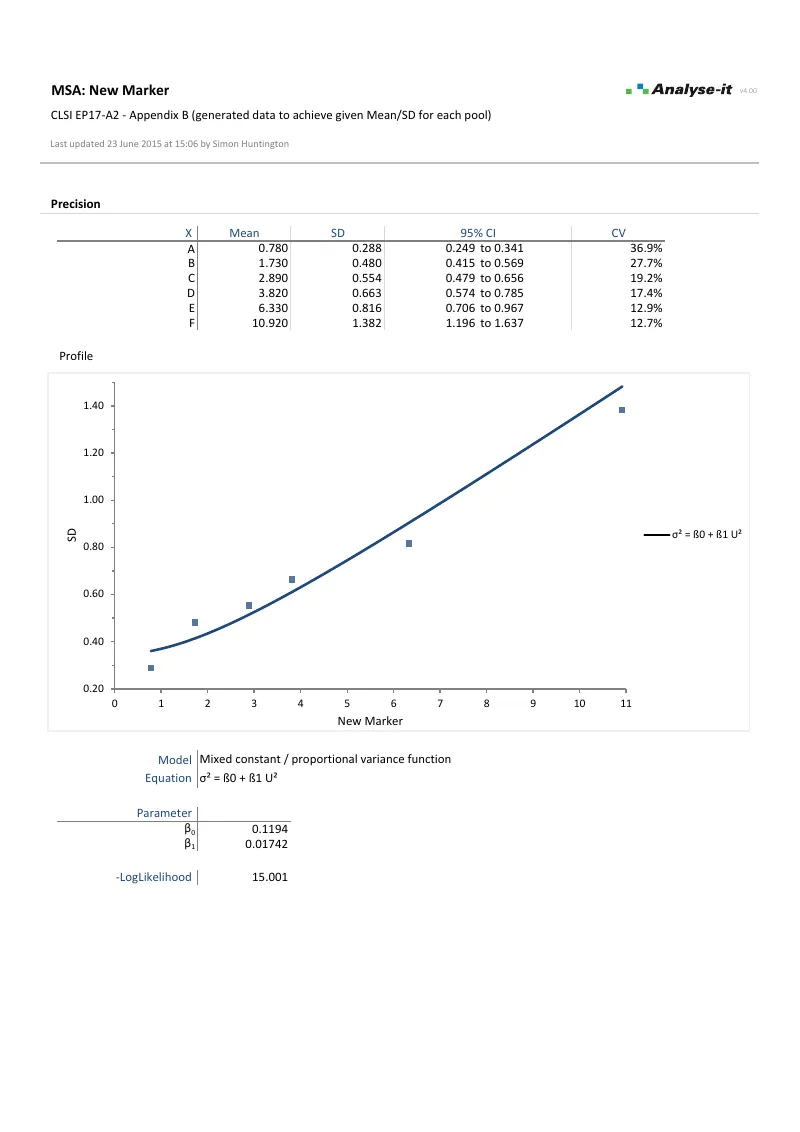
LoQ directly from the precision profile variance function, linking EP17-A2 to EP05-A3 precision data — the concentration where imprecision drops below the threshold for clinically reliable quantitative results, such as a CV of 10% or 20%.
Example analyses
See detection capability results in detail — LoB, LoD, probit regression, and LoQ — using CLSI example datasets you can download and follow along with.
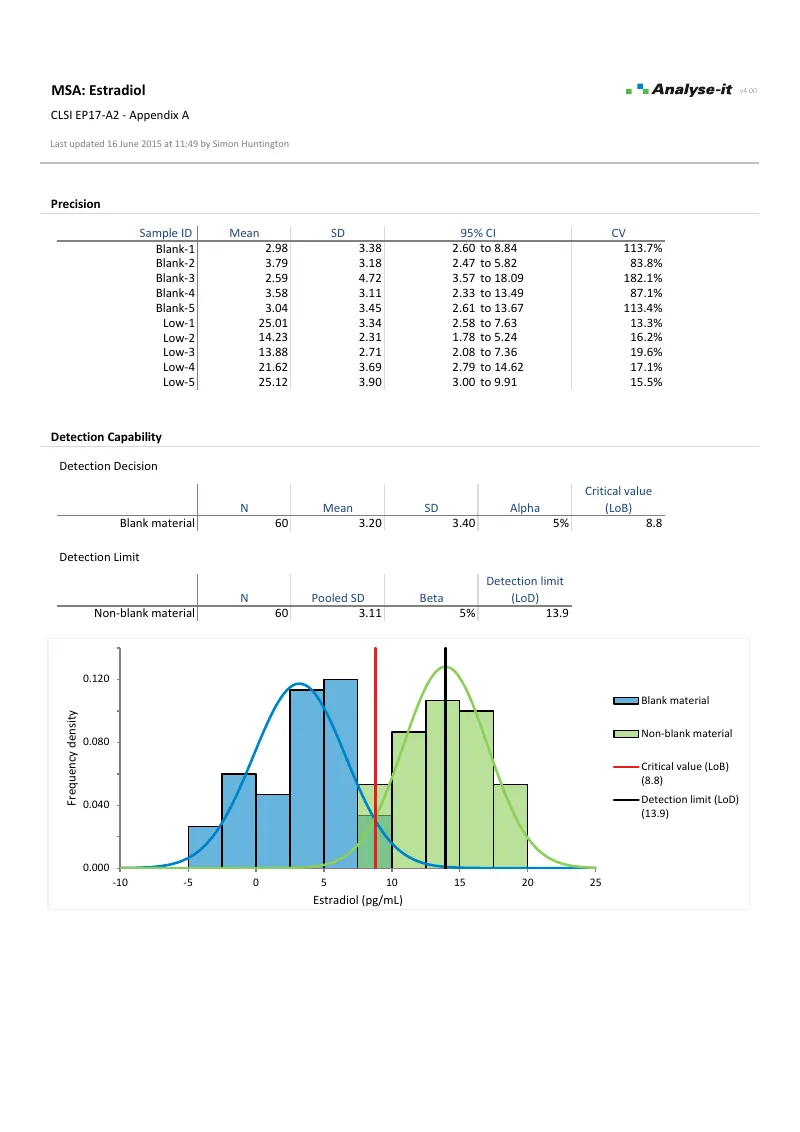 EP17-A2 — Appendix A
EP17-A2 — Appendix AEstradiol detection capability. Parametric LoB from 5 blank samples and LoD from pooled SD of 5 low-level samples. Frequency density histogram with LoB and LoD lines.
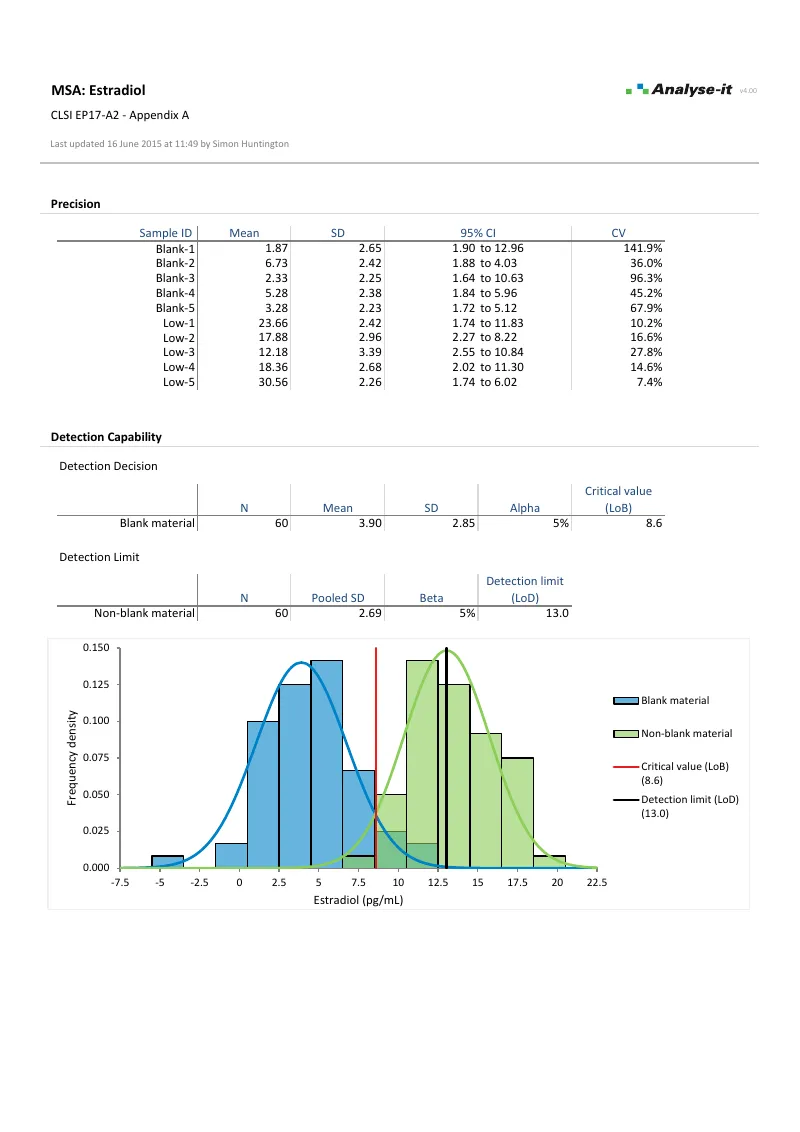 EP17-A2 — Appendix A
EP17-A2 — Appendix AEstradiol detection capability, second reagent lot. Same structure as Example 1 — parametric LoB and LoD with frequency density histogram. Second reagent lot.
Part of measurement system analysis
Detection capability is one part of measurement system analysis, alongside precision (EP05-A3), linearity (EP06-A), bias/trueness verification (EP15-A3), and interference evaluation (EP10-A3-AMD). Precision profiles from EP05-A3 feed directly into detection capability estimation.
See the full MSA workflow on the measurement system analysis page →
Software you can trust
Technical details
CLSI protocols
- EP17-A2: Evaluation of Detection Capability for Clinical Laboratory Measurement Procedures
Limit of blank (LoB)
- Parametric (SD of blank material) new in v4.00
- Non-parametric (quantile of blank material) new in v4.00
- From precision profile variance function new in v4.00
Limit of detection (LoD)
- Pooled SD of non-blank materials new in v4.00
- From precision profile variance function new in v4.00
- Probit regression new in v5.50
Limit of quantitation (LoQ)
- From precision profile variance function new in v4.00
Plots
- Frequency density histogram with LoB and LoD
- Probit regression curve new in v5.50