Method validation software for clinical laboratories and IVD manufacturers 11 CLSI protocols using current guideline versions in a single validated package — precision, linearity, detection capability, method comparison, reference intervals, and diagnostic performance.
Trusted by 75,000 scientists at most of the top-10 IVD manufacturers, and at thousands of ISO 15189, ISO/IEC 17025, and CLIA-regulated laboratories worldwide for over 30 years.






11 CLSI protocols, one package
EP05-A3, EP06-A, EP09-A3, EP10-A3-AMD, EP12-A2, EP14-A3, EP15-A3, EP17-A2, EP21-A, EP24-A2, and EP28-A3C. Current guideline versions, not older editions. One tool for the entire validation workflow — no gaps to fill with spreadsheets or secondary software.
Validated against CLSI reference data
Every calculation runs in Analyse-it's own engine — no Excel formulas, no third-party functions. Results are validated against the example datasets published in the CLSI guidelines before every release. Defensible in a 510(k), CE-IVD technical file, CAP inspection, or ISO 15189 audit.
Data stays in your facility
Analyse-it runs entirely within Microsoft Excel on your PC. No cloud processing, no data transmission — your pre-submission and patient-adjacent data stays within your facility under your data governance controls.
Method validation covers a lot of ground. IVD manufacturers need to establish precision, linearity, bias, and detection capability during development, demonstrate method comparability with a predicate, establish reference intervals and diagnostic accuracy for product labelling, and produce the statistical evidence for a 510(k) submission or CE-IVD technical file. Clinical laboratories need to verify that a new analyser or reagent system meets manufacturer claims before it goes into clinical use, establish or verify reference intervals for their patient population, and demonstrate compliance during ISO 15189, CLIA, or CAP inspections. Both audiences need the same statistical rigour — the difference is regulatory context, not analytical depth.
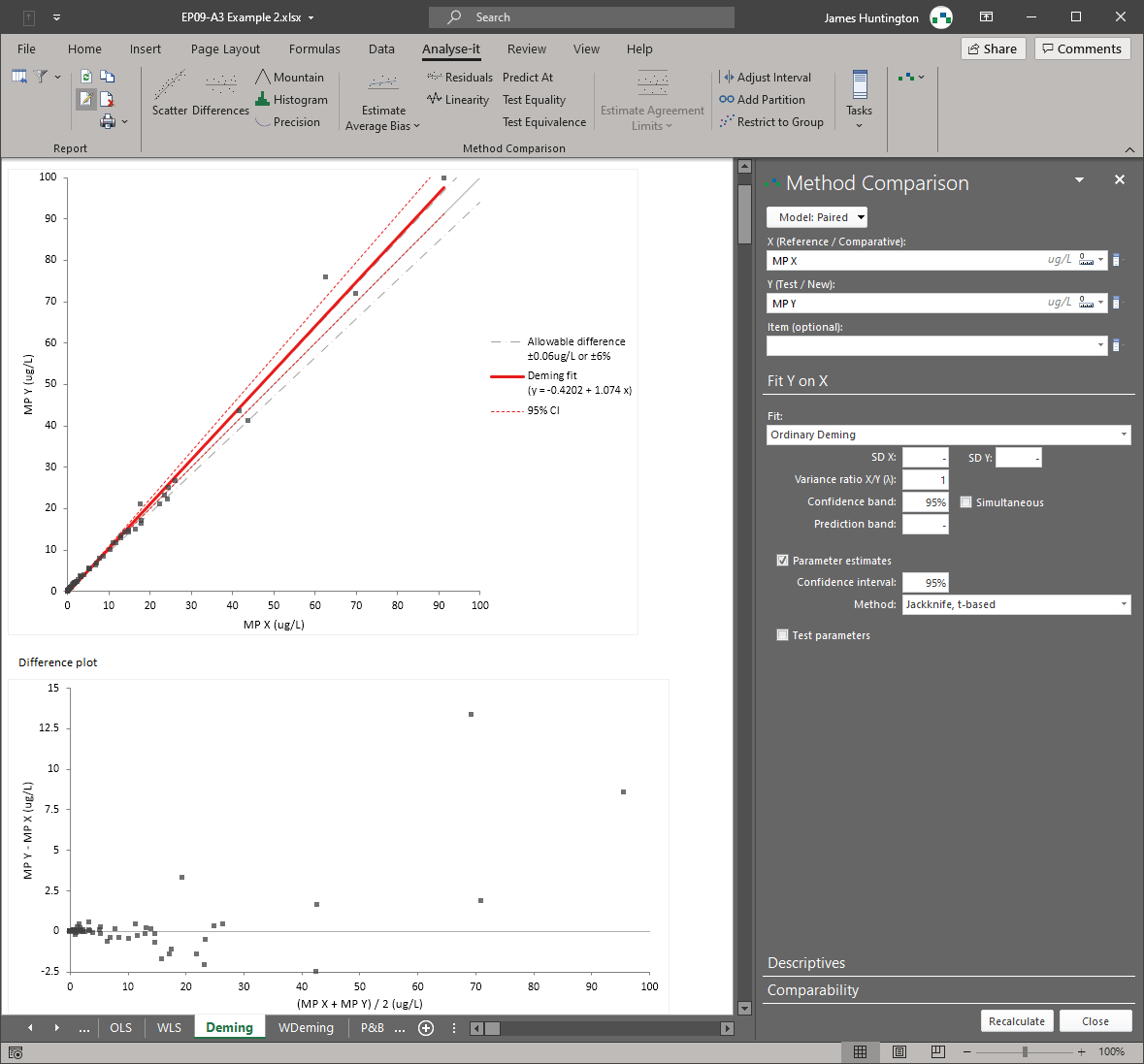
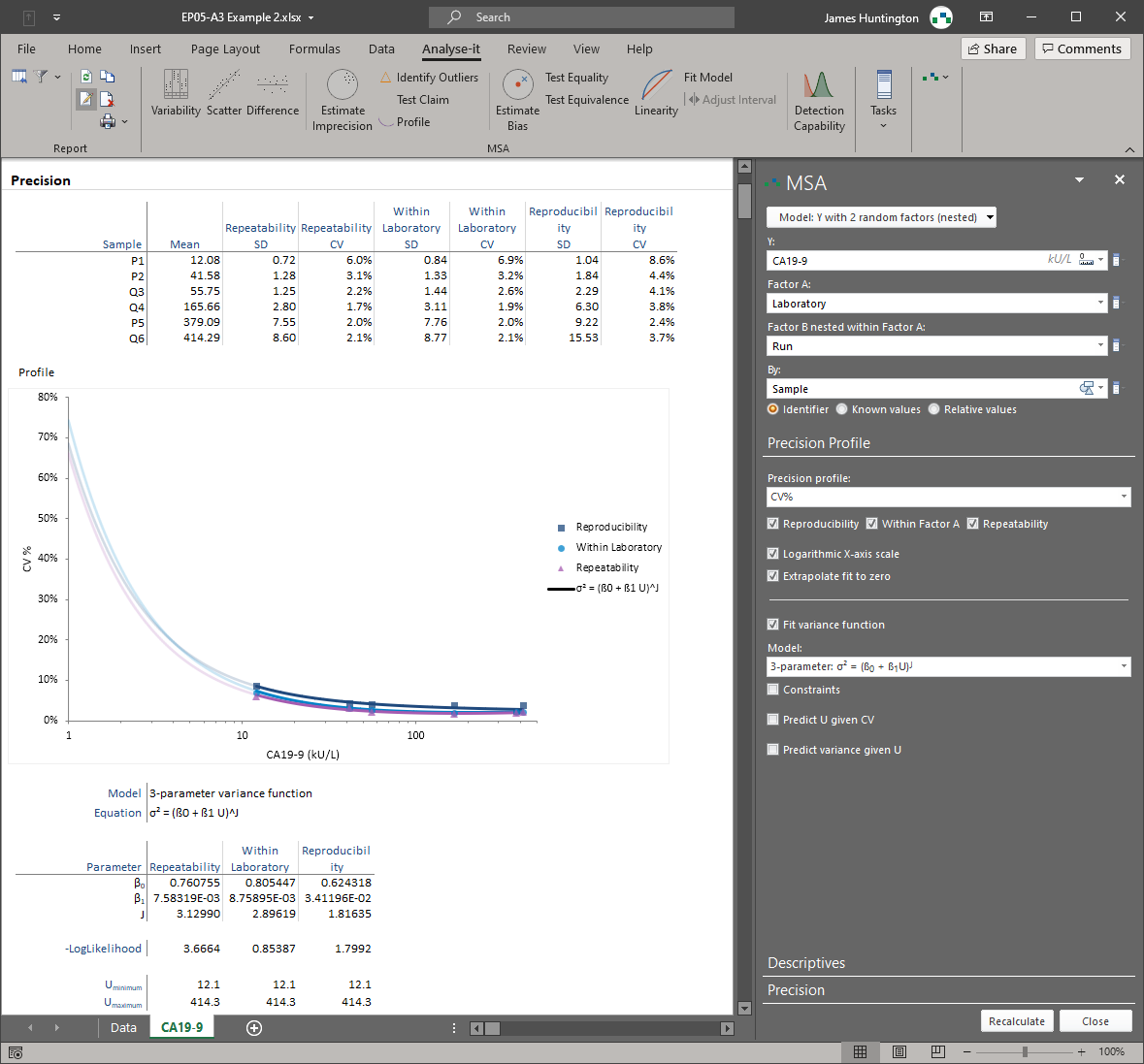
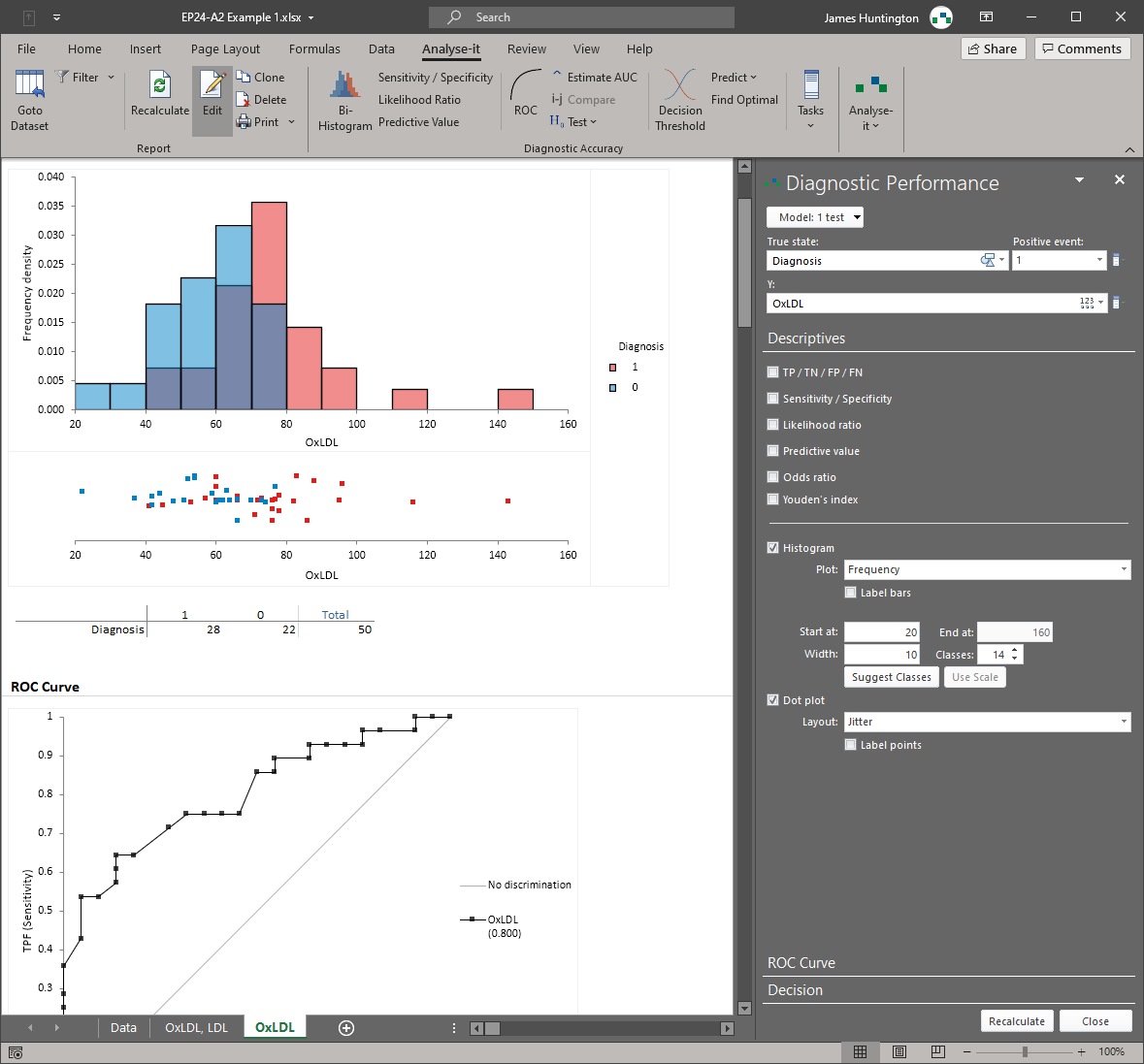
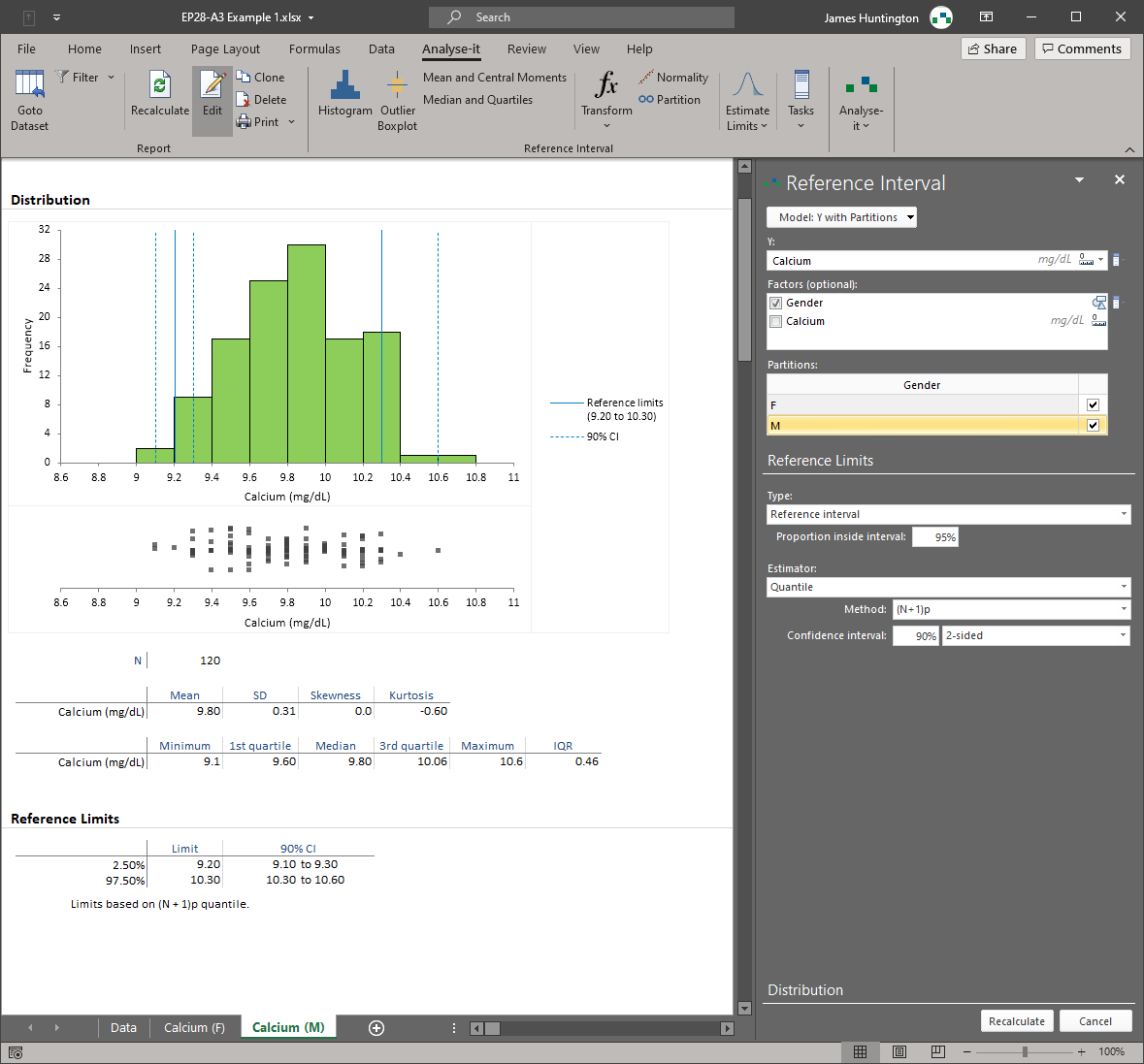
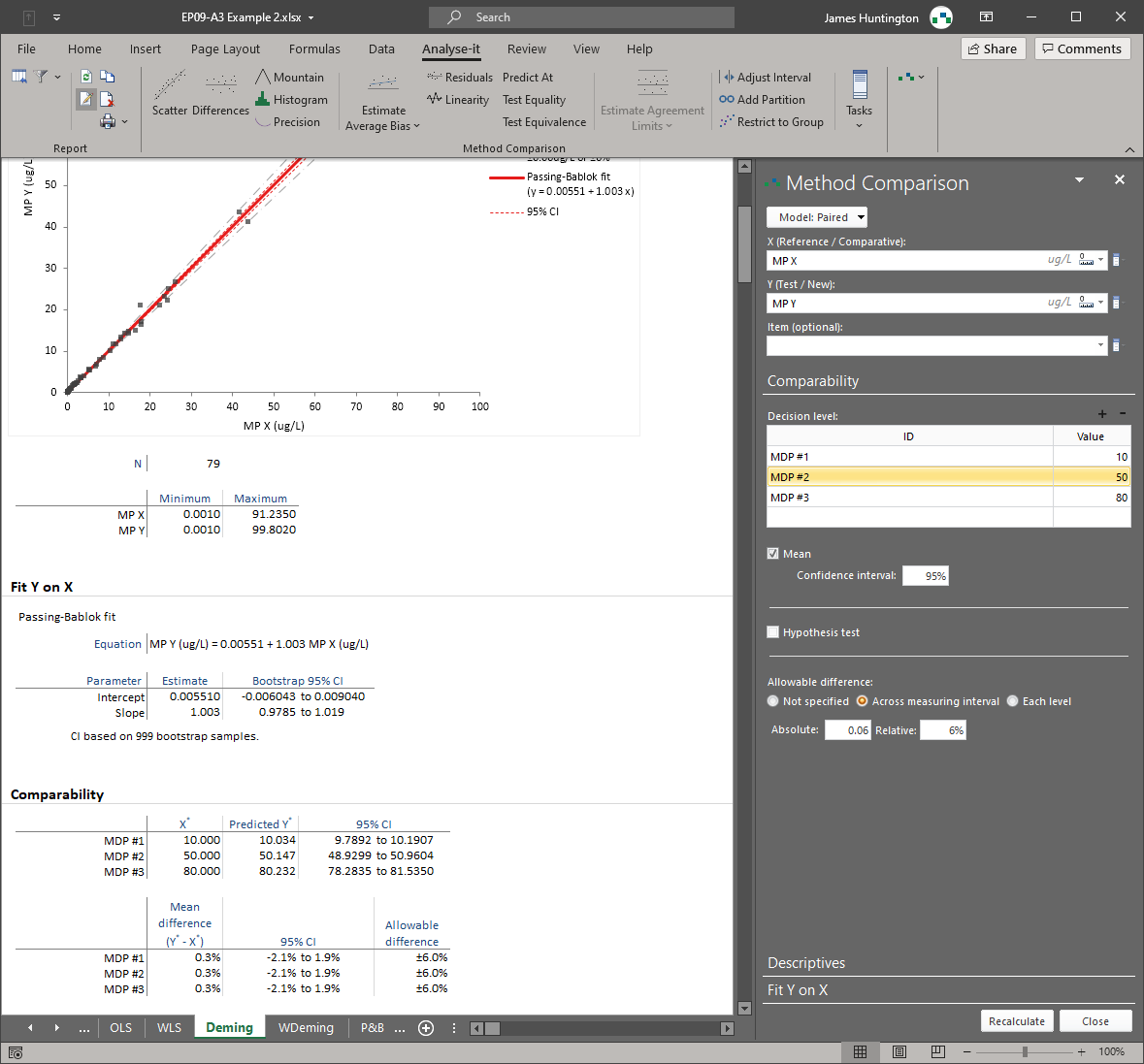
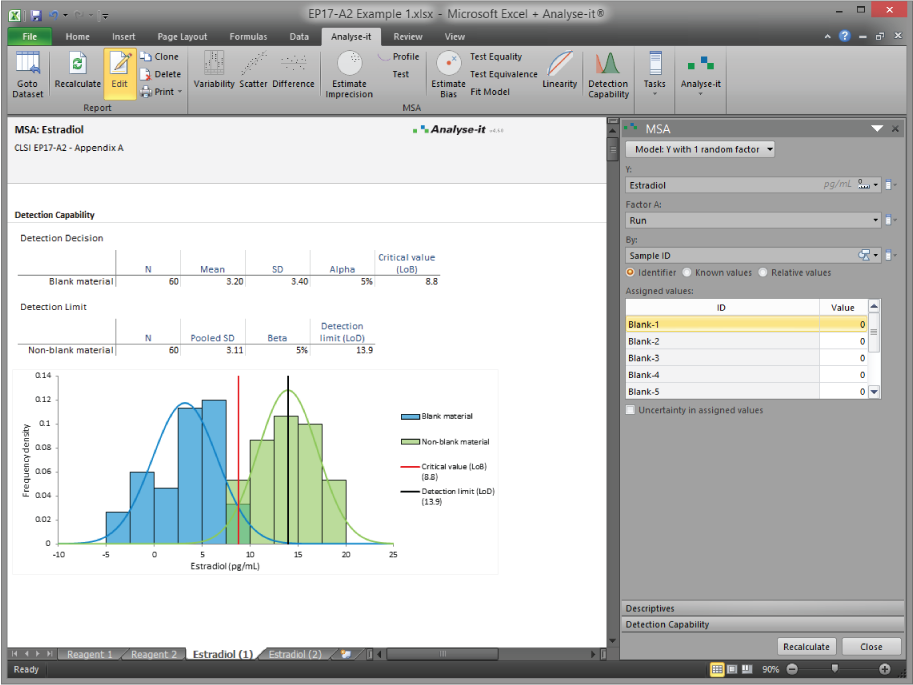
Analyse-it implements 11 CLSI protocols using current guideline versions. Precision studies with flexible nested designs per EP05-A3. Linearity evaluation per EP06-A. Interference testing per EP10-A3-AMD. Verification against manufacturer claims per EP15-A3. Detection capability — LoB, LoD, and LoQ — per EP17-A2. Method comparison using Passing-Bablok, Deming, Weighted Deming, OLS, Weighted OLS, and Bland-Altman per EP09-A3, with bias at clinical decision points, replicate support, interval partitioning, and total analytical error estimation per EP21-A. Reference intervals using parametric, non-parametric, robust, bootstrap, and Harrell-Davis methods per EP28-A3C, with partitioning and transfer/verification. Diagnostic performance with ROC curves, AUC comparison, and qualitative test evaluation per EP24-A2 and EP12-A2.
The practical value of having all of this in one package is not just convenience — it is continuity. A method validation project moves through measurement system characterisation, method comparison, reference interval establishment, and diagnostic performance evaluation. Each stage informs the next. Having the entire workflow in one tool, on the same data, in the same workbook means the results are consistent, the audit trail is clear, and the output can go directly to regulatory affairs or the accreditation file without reformatting or reconstruction.
Scientists at Abbott, Roche, Thermo Fisher, Siemens Healthineers, and most of the world's leading IVD manufacturers have relied on Analyse-it for over 30 years. So have clinical laboratories running verification studies, reference interval programmes, and method changeovers at hospitals and reference laboratories worldwide.
Seen in the field
What's included
-
Characterise measurement system performance
Precision with flexible nested variance component designs (EP05-A3). Linearity with polynomial regression and nonlinearity testing (EP06-A). Verification against manufacturer claims (EP15-A3). Detection capability — LoB, LoD, and LoQ (EP17-A2). Interference testing (EP10-A3-AMD). See measurement system analysis details.
-
Compare methods and estimate bias
Passing-Bablok, Deming, Weighted Deming, OLS, Weighted OLS regression, and Bland-Altman limits of agreement — all six methods in a single analysis per EP09-A3. Bias at clinical decision points, replicate support, interval partitioning, and total analytical error estimation per EP21-A. See method comparison details.
-
Establish and verify reference intervals
Parametric, non-parametric, robust, bootstrap, and Harrell-Davis quantile methods per EP28-A3C. Partition by sex, age, ethnicity, or any factor. Full range of transformations for non-normal data. Transfer and verify existing intervals against a new procedure or population. See reference interval details.
-
Evaluate diagnostic test accuracy
ROC curves with DeLong AUC comparison for quantitative tests (EP24-A2). Sensitivity, specificity, likelihood ratios, predictive values, optimal threshold determination. Qualitative test evaluation (EP12-A2). Test comparison for up to 10 paired or independent tests. See diagnostic performance details.
-
Validated engine you can defend at inspection
Every calculation runs in Analyse-it's own engine — no Excel formulas or third-party functions. Validated against the CLSI published example datasets before every release. Results go directly into a regulatory dossier, validation report, or accreditation file. See the development and validation process.
Trusted by 75,000 scientists at most of the top-10 IVD manufacturers, and at thousands of ISO 15189, ISO/IEC 17025, and CLIA-regulated laboratories worldwide for over 30 years.
Try free for 15 days Read more...
Read more...