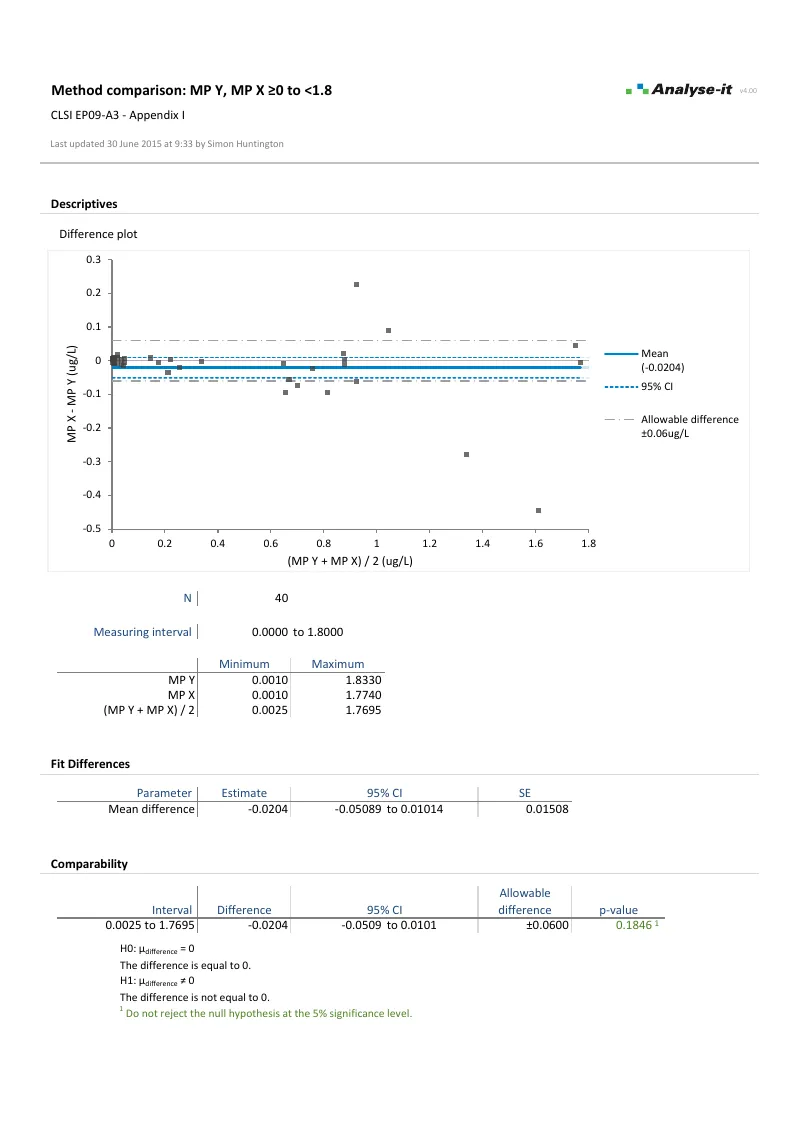
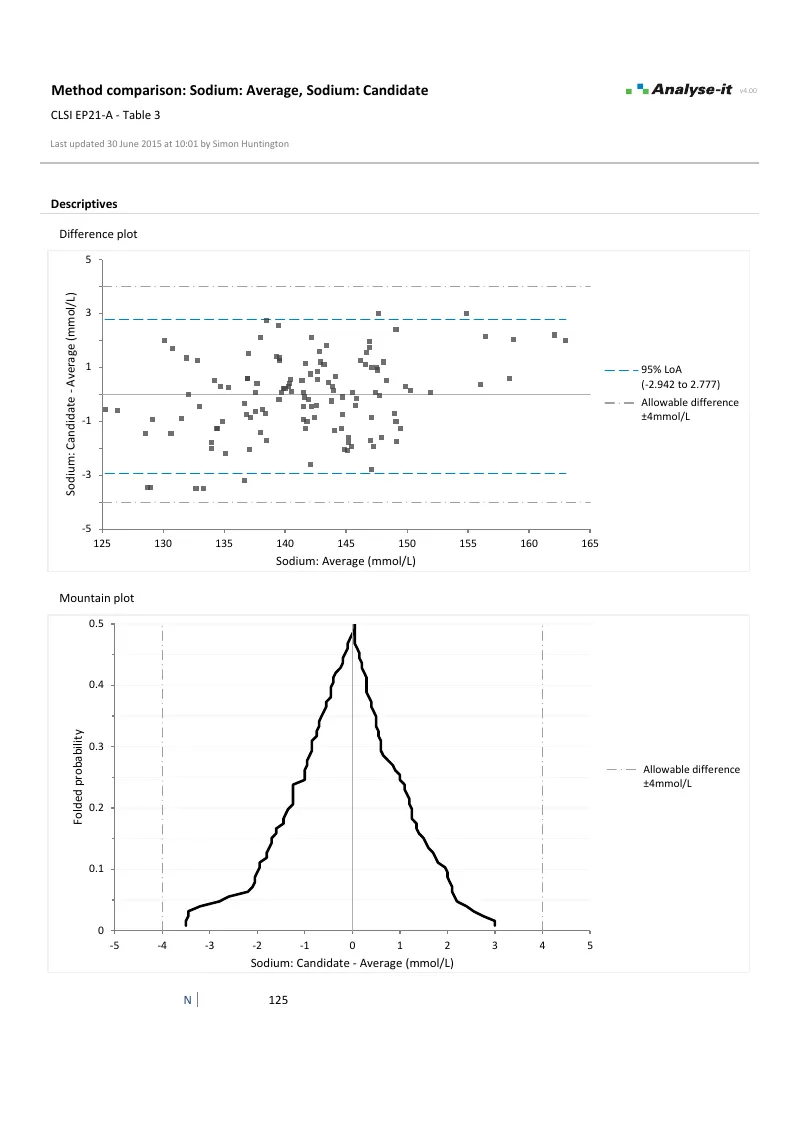
Bland-Altman analysis for method agreement Limits of agreement with constant and non-constant precision, mean and median bias, replicate measurements, and confidence intervals on the limits — per EP09-A3.
See the agreement between methods at every concentration
A regression line tells you the average relationship between two methods. It doesn’t show you the individual differences, whether they’re constant or widen at higher concentrations, or whether any single patient result might be clinically misclassified. The difference plot shows all of that — which is why Bland-Altman has become the standard approach for visualising and quantifying method agreement across clinical laboratories, IVD development, and published research.
Analyse-it provides the full Bland-Altman implementation with three bias models, limits that adapt to the precision structure of your data, and confidence intervals on the limits themselves — not just on the bias estimate. The mountain plot alongside gives you the cumulative distribution of differences for a second perspective on agreement.
Principal Scientist, Product Development
Biosite / Inverness Medical Innovations
What's included
Difference plot with limits of agreement and allowable difference band
Differences plotted against the best estimate of the true value, with bias, limits of agreement, and allowable difference band overlaid. Specify allowable difference as absolute, percentage, or combination — such as “±4 mmol/L” or “10%, with a minimum of 5 mg/dL” — to assess whether agreement is within clinically acceptable limits. Plot as difference, relative difference, or ratio. Mountain plot alongside for the cumulative distribution of differences, also with allowable difference band.Mean, median, and linear fit bias
Three bias models in one analysis. Mean bias for centred, symmetric differences. Median bias for skewed distributions. Linear fit when bias scales with concentration. Compare all three on the same data and choose the one that represents the relationship.Constant and non-constant precision limits
Horizontal limits of agreement for constant precision across the range. V-shaped limits that widen proportionally when precision is non-constant. Nonparametric limits when differences are non-normally distributed.Confidence intervals on the limits themselves
Confidence intervals on both the bias estimate and the upper and lower limits of agreement — reporting the uncertainty in the agreement boundary, not just the central estimate.Partition the measuring range
Partition the data into separate measuring intervals, each with its own bias estimate, limits of agreement, and allowable difference assessment — for example, ±0.06 μg/L below the clinical threshold and ±6% above it. Use any fit within each interval per EP09-A3.Replicate measurements with correct variance
Singlicate, duplicate, or any number of replicates. Within-subject variation estimated directly from the replicate structure and limits adjusted accordingly — not approximated by averaging.
Example analyses
See Bland-Altman results in detail — difference plots, limits of agreement, mountain plots, and allowable difference bands — using CLSI example datasets you can download and follow along with.
Part of the method comparison workflow
Bland-Altman is one of six methods in the method comparison analysis. For a regression-based bias estimate, see Passing-Bablok for a non-parametric approach or Deming and Weighted Deming for a parametric approach.
Software you can trust
Technical details
CLSI protocols
- EP09-A3: Measurement Procedure Comparison and Bias Estimation Using Patient Samples
Bland-Altman agreement
- Mean or Median bias for constant precision (horizontal limits)
- Linear regression fit bias new in v3.75 for non-constant precision (V-shaped limits)
- Limits of agreement with confidence intervals
Study design
- Singlicate, duplicate, and replicate measurements
- Reduce or partition measuring interval new in v4.00
Plots
- Difference plot with bias, limits of agreement and allowable difference
- Plot difference, relative difference, or ratio
- Histogram of differences
- Mountain plot with allowable difference band new in v3.71