Bland–Altman agreement software for method comparison Limits of agreement with mean, median, and linear fit bias, constant and non-constant precision, mountain plots, replicate handling, and qualitative agreement measures — the standard method comparison analysis for clinical research.
See the agreement between two measurement methods
When you introduce a new analyser, switch reagent systems, or compare a point-of-care device against a laboratory method, you need to know whether the two methods agree well enough for clinical use. Correlation tells you the methods are related — it does not tell you they agree. A Bland–Altman difference plot shows you the bias between methods, how it varies across the measuring range, and where the limits of agreement fall relative to what is clinically acceptable.
The full Bland–Altman analysis. Mean and median bias with confidence intervals. Limits of agreement for constant precision and non-constant precision that widens across the range. Linear fit on the difference plot to detect concentration-dependent bias. Mountain plots as an alternative visualisation. Support for replicate measurements with proper within-subject variance estimation. Qualitative agreement with PPA/NPA and kappa for binary method comparisons.
Medical Technologist QA/Ancillary Testing
Veterans Administration
What's included
Quantify agreement with mean bias and limits of agreement
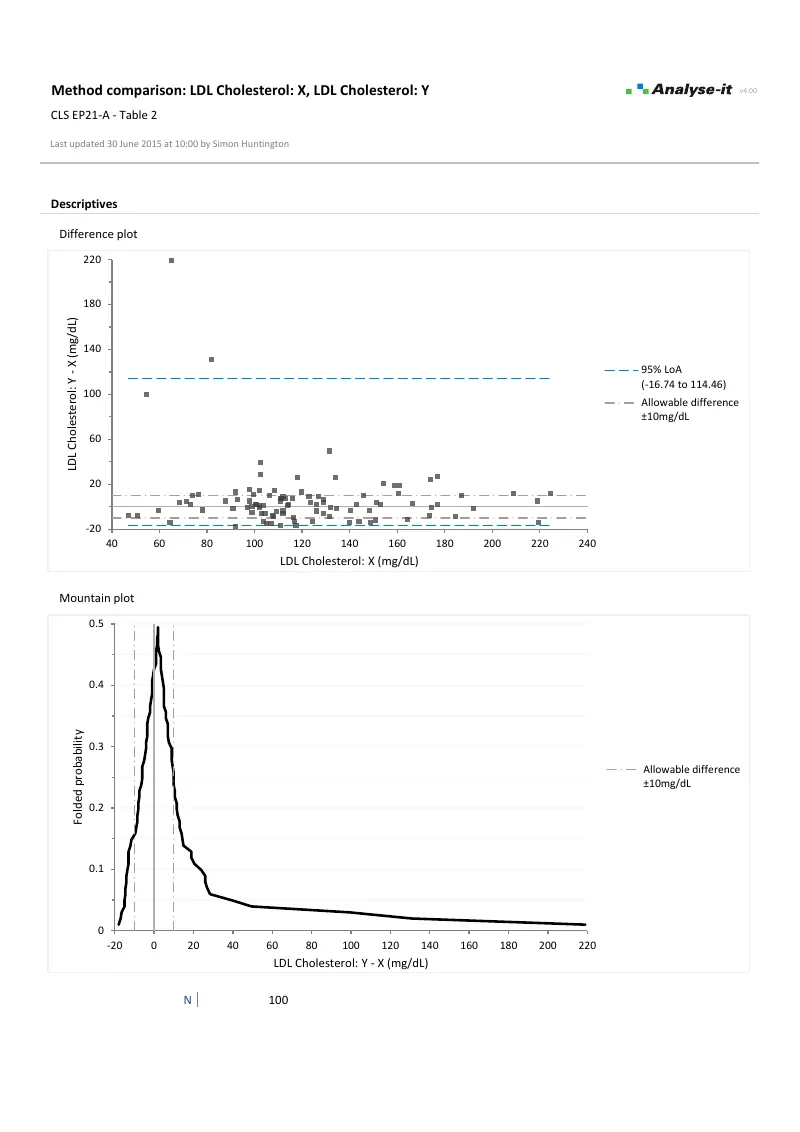
Mean bias and median bias with confidence intervals. 95% limits of agreement show the range within which differences between the two methods are expected to fall. Overlay an allowable difference specification — such as ±4 mmol/L for glucose or ±10 mg/dL for LDL cholesterol — to see immediately whether agreement meets clinical requirements. Difference plot with histogram of differences.Detect concentration-dependent bias with linear fit on the difference plot
Linear fit on the difference plot to quantify proportional bias that changes across the measuring range. Separate constant bias (intercept) from concentration-dependent bias (slope) and determine whether either is clinically meaningful.Handle non-constant precision with regression-based limits of agreement
Regression-based (V-shaped) limits of agreement for data where precision is non-constant across the measuring range. The limits widen or narrow with concentration rather than assuming the same variability everywhere.Visualise the difference distribution with mountain plots
The mountain plot (folded empirical CDF) shows the entire distribution of differences between methods in a single curve. Overlay an allowable difference band to see the proportion of differences that fall outside clinical requirements. A useful complement to the difference plot, especially when presenting to audiences unfamiliar with limits of agreement.Estimate within-subject variance from replicate measurements
Handle singlicate, duplicate, and replicate measurements correctly. Within-subject variation is properly estimated from replicates and confidence intervals adjusted accordingly — not approximated by averaging replicates before analysis.Evaluate qualitative agreement with PPA/NPA and kappa
When comparing two methods with binary outcomes — positive or negative — proportion in positive agreement (PPA) and negative agreement (NPA) with Wilson score confidence intervals. Kappa and weighted kappa for chance-corrected agreement. Kappa test for agreement beyond chance.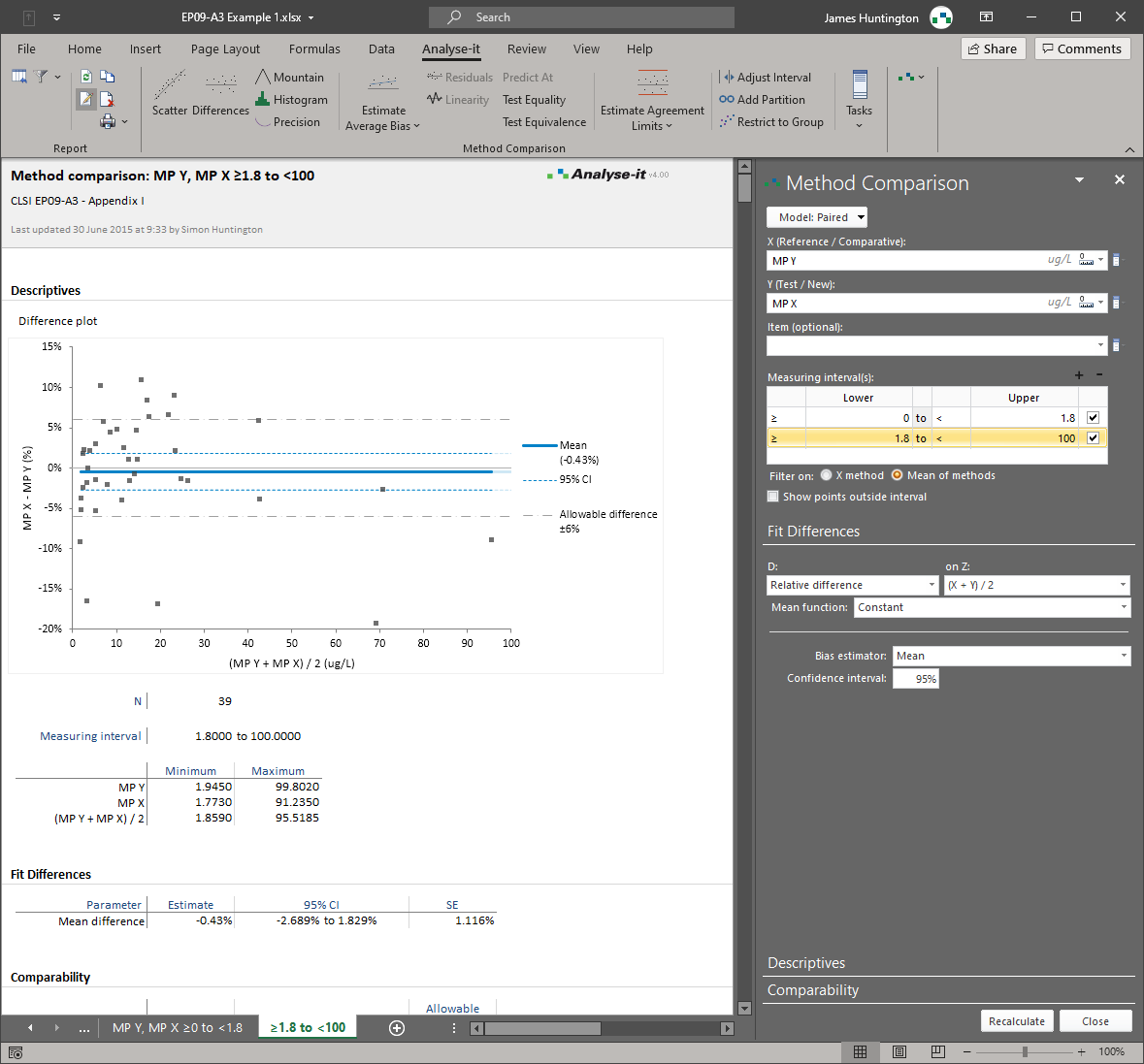
Example analyses
See Bland–Altman agreement results in detail — difference plots, limits of agreement, and mountain plots — using example datasets you can download and follow along with.
Part of the Medical edition
Bland–Altman agreement is one part of the Medical edition, alongside diagnostic accuracy, reference intervals, and survival analysis — plus the full Standard edition for hypothesis testing, regression, and descriptive statistics.
For formal method validation with Passing–Bablok, Deming regression, bias at clinical decision points, and CLSI protocol support, see the Method Validation edition.
Software you can trust
Technical details
Bland–Altman agreement
- Mean bias with confidence interval
- Median bias with confidence interval
- Linear fit for concentration-dependent bias new in v3.75
- Limits of agreement — constant precision (horizontal)
- Limits of agreement — non-constant precision (regression-based) new in v3.75
- Confidence intervals on limits of agreement
- Allowable difference specification and overlay
- Singlicate, duplicate, and replicate measurements
- Within-subject variance estimation from replicates
- Precision (SD or CV) for each method
- Pearson r correlation coefficient
Qualitative agreement
- Proportion in positive/negative agreement (PPA/NPA) with Clopper–Pearson exact or Wilson score CI
- Kappa and weighted kappa with Wald Z CI
- Kappa test for agreement
Plots
- Difference / relative difference / ratio plot against X or mean of methods with allowable difference band and histogram
- Mountain plot with allowable difference band new in v3.71
- Scatter plot with identity line
- Vary colour of points by a factor