Reference interval software for clinical laboratories Five quantile methods including robust and bootstrap approaches, partitioning by sex, age, or ethnicity, full range of transformations, outlier screening, normality assessment, and transfer and verification of existing intervals.
Establish the reference ranges clinicians depend on
Every laboratory result a clinician sees is interpreted against a reference interval. If the interval is wrong — too wide, too narrow, derived from the wrong population, or not partitioned where it should be — normal patients get flagged and abnormal patients get missed. Establishing defensible reference intervals from your own population, or verifying that a manufacturer’s intervals apply to your patients, is one of the most consequential statistical tasks a laboratory performs.
Five quantile methods to match your sample size and distribution. Partition by any factor when subgroups need separate ranges. Screen outliers, assess normality, apply transformations when the raw distribution doesn’t fit. Transfer existing intervals to a new method using regression, or verify them with a binomial test.
Director, Clinical Development
Johnson and Johnson
What's included
Choose from five quantile methods to match your sample size and distribution
Parametric quantiles when the distribution is Gaussian. Non-parametric with three computation approaches — (N+1)p, Np+½, (N+⅓)p+⅓ — when you have 120+ observations. Robust bi-weight for small or moderately skewed samples. Bootstrap for distribution-free estimation. Harrell–Davis for a smooth quantile estimate. Confidence intervals on all reference limits.Partition by factors that require separate reference ranges
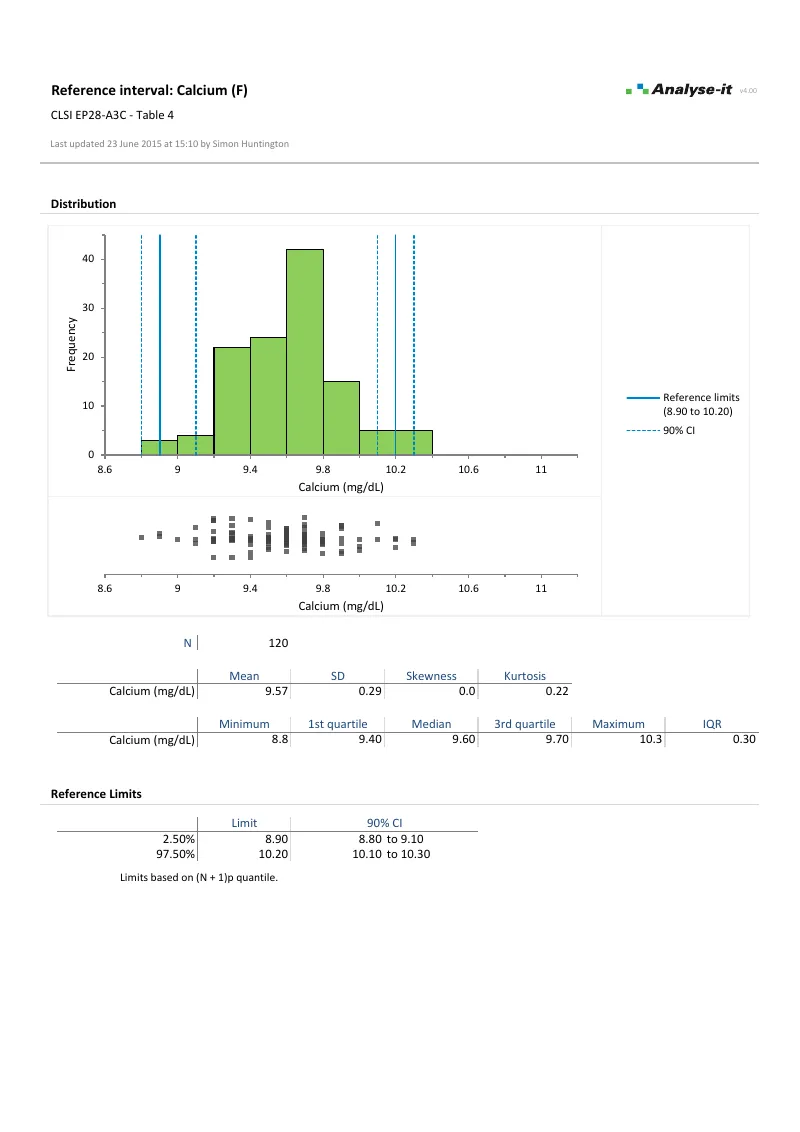
Partition by sex, age group, ethnicity, or any combination of factors — such as separate calcium intervals for males and females, or age-stratified alkaline phosphatase ranges for paediatric populations. Outlier screening and normality assessment run independently for each subgroup.Screen outliers and assess normality before establishing limits
Tukey box plots for outlier identification. Shapiro–Wilk and Anderson–Darling normality tests. Frequency histogram with normal overlay. Normal Q–Q plot with Lilliefors confidence band.Apply transformations when the raw distribution doesn’t fit
Log, square root, cube root, Box–Cox, Manly exponential, and two-stage exponential/modulus transformations. Choose the transformation that achieves the best fit, compute parametric reference limits on the transformed scale, and back-transform to original units. Reciprocal and cube root also available.Transfer existing intervals or verify them against your patient population
Transfer a reference interval from one measurement procedure to another using the regression function from a method comparison study. Verify that a manufacturer’s published interval applies to your patient population with a binomial test — such as confirming that at least 90% of 20 reference individuals fall inside the claimed interval.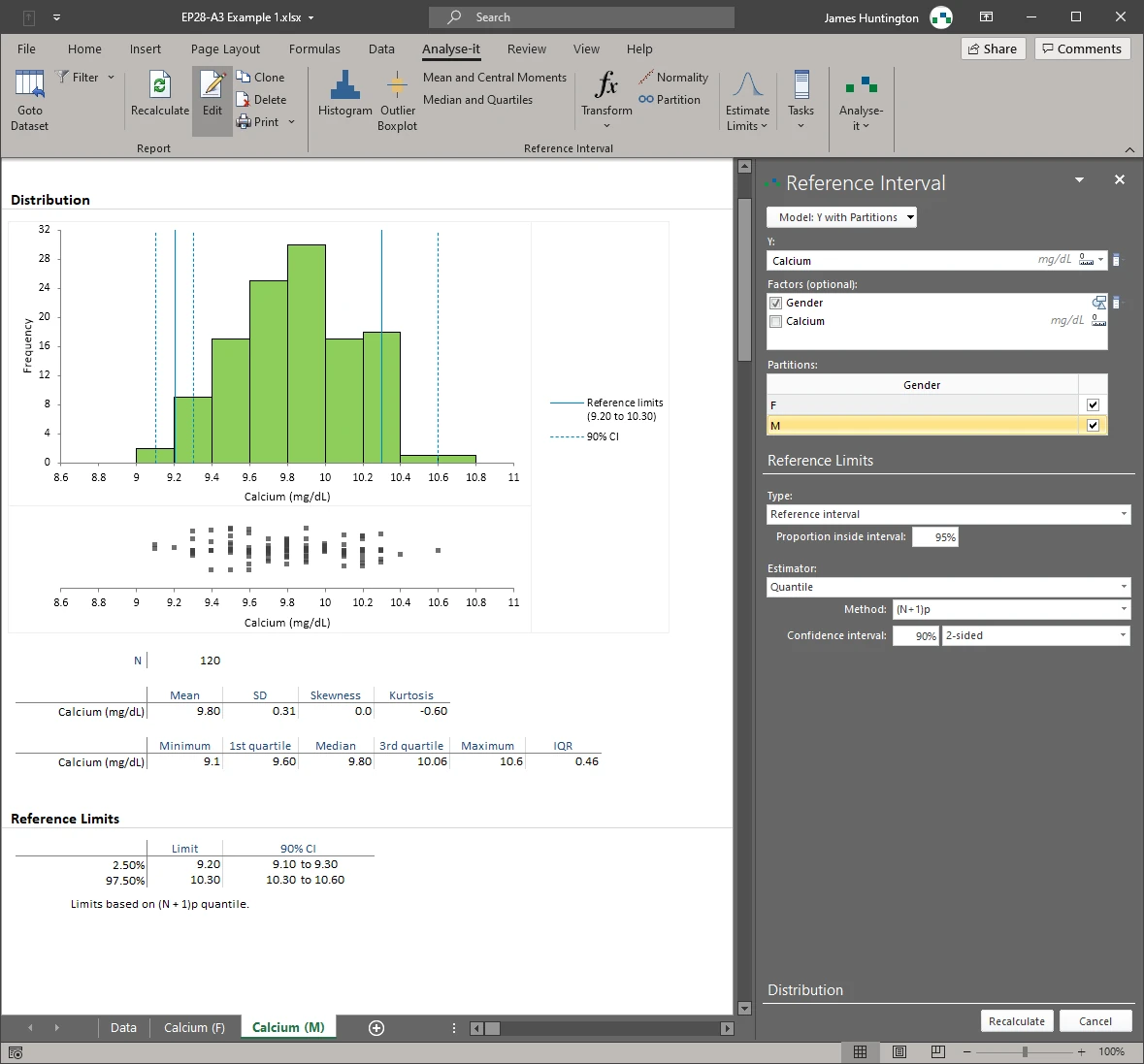
Example analyses
See reference interval results in detail — partitioned intervals, histograms with reference limits, and outlier screening — using example datasets you can download and follow along with.
Part of the Medical edition
Reference intervals are one part of the Medical edition, alongside diagnostic accuracy, Bland–Altman agreement, and survival analysis — plus the full Standard edition for hypothesis testing, regression, and descriptive statistics.
For formal method validation with CLSI EP28-A3C protocol support, see the Method Validation edition.
Software you can trust
Technical details
Establish reference limits
- Normal (parametric) quantile
- Non-parametric quantile: (N+1)p, Np+½, (N+⅓)p+⅓
- Harrell–Davis quantile
- Bootstrap quantile
- Robust bi-weight quantile for symmetric and skewed small samples
- Confidence intervals on all reference limits
Transfer / verify
- Transfer existing reference interval using method comparison regression function
- Binomial test for proportion inside reference interval
Partitioning & transformations
- Partition by factor(s) new in v4.00
- Reciprocal transformation
- Log transformation
- Square and cube root
- Box–Cox new in v3.52
- Manly exponential new in v4.00
- Two-stage exponential / modulus new in v4.00
Normality testing
- Shapiro–Wilk test
- Anderson–Darling test
- Normal Q–Q plot with Lilliefors confidence band
Plots
- Frequency distribution histogram with normal overlay and reference limits
- Tukey outlier box plot
- Normal Q–Q plot with Lilliefors confidence band