Estimating precision for a multi-site study
Estimate the precision of the measurement procedure at multiple sites and samples.
-
Open the file tutorials\EP05-A3 Appendix B.xlsx.
The worksheet opens showing 4 columns. The Sample column identifies the levels of the analyte in the sample (6 levels). Laboratory identifies the site (3 laboratories), Run identifies the run (5 runs per site). CA19-9 (kU/L) identifies the measured value for 5 replicates of sample in each run.
- Click a cell in the dataset.
-
On the Analyse-it ribbon tab, in the
Statistical Analyses group, click Precision, and then click 2 Factor Nested.
The analysis task pane opens.
- In the Y drop-down list, select CA-19.
- In the Factor A drop-down list, select Laboratory.
- In the Factor B nested within Factor A drop-down list, select Run.
- In the By drop-down list, select Sample.
- In the Estimator drop-down list, select SD.
- In the Confidence Interval edit box, type 95%, and then in the Method drop-down list, select Exact / Satterthwaite.
- In the Conditions drop-down list, select Reproducibility / Repeatability to label the innermost variance component repeatability and the outermost reproducibility.
- On the Analyse-it ribbon tab, in the MSA group, click Variability plot.
- On the Analyse-it ribbon tab, in the MSA group, click Identify Outliers, and then on the analysis task panel in the Granularity drop-down list, select Level, Factor A so that outliers are detected for each sample and laboratory.
-
Click Calculate.
The analysis report opens.
The variability plots show a simple visual assessment of the closeness of agreement between the measured quantity values. The purple lines show the mean of each run, the light blue lines show the mean of each laboratory, and the dark blue line the overall grand mean.
You should observe the scatter of the points to ensure there are no obvious problems. No individual measurements stand out as highly aberrant relative to the bulk of the data and none of the plots exhibit any apparent drift capable of distorting the results.
The abbreviated variance components table shows the required precision statistics expressed numerically as the standard deviation (SD) and coefficient of variation (CV).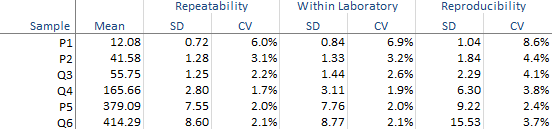
The detailed variance components table show the precision expressed numerically as the chosen measure of imprecision along with a confidence interval for each component.